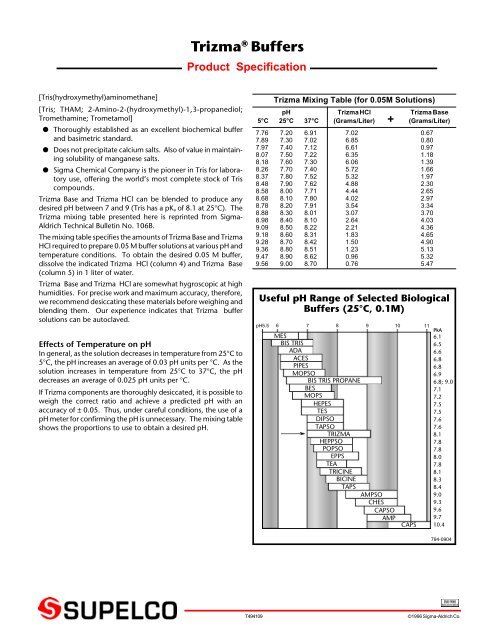
SOLVED: SDS-PAGE 10X gel running buffer 248 mM Tris (Trizma base) (121.14 g/mol) 1.92 M glycine (75.07 g/mol) 1% w/v SDS (288.38 g/mol)

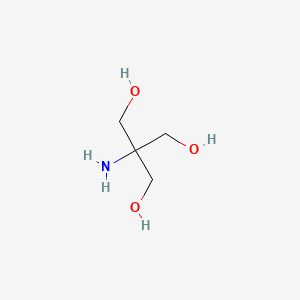
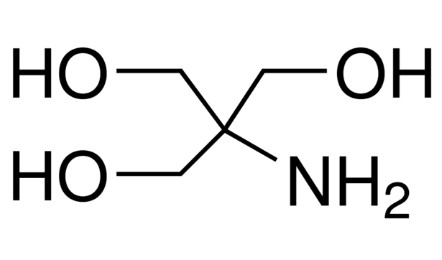
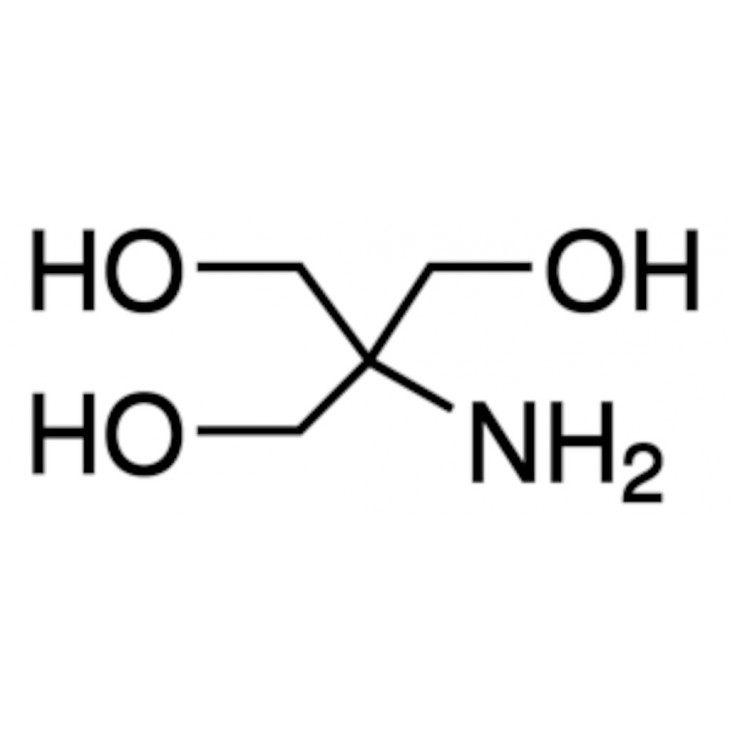
SOLVED: Tris(hydroxymethyl)aminomethane, abbreviated as Tris or TRIZMA, is frequently used to make buffers in biochemistry laboratories. Tris has a molecular weight of 121.1 g/mol, and its solution in water is basic with










![T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams T60040-500.0 - Tris Base Ultra Pure [Tris (Hydroxymethyl) Aminomethane], 500 Grams](https://d2gdaxkudte5p.cloudfront.net/system/images/T60040-500.0_.jpg)