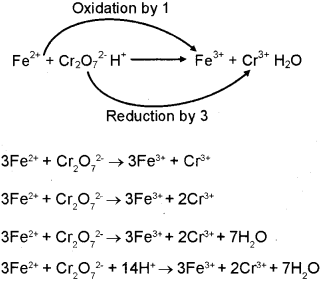
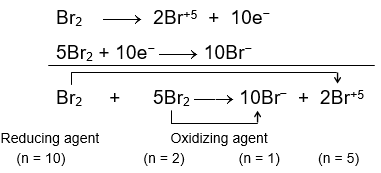
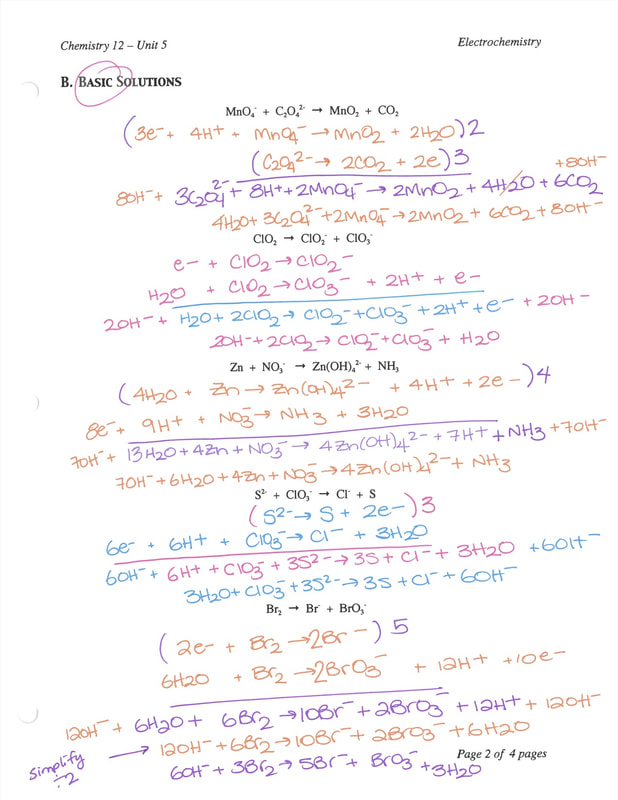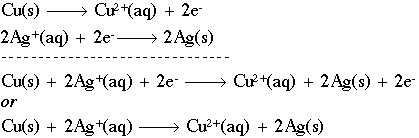Oxidation and Reduction reactions- Definition, Reactions, Examples, Balancing the oxidation and reduction reactions, Videos and FAQs

Write balanced equations for the half reactions and calculate the reduction potentials at 25^∘C for the following half cells:(a) Cl^-(1.2M) | Cl1(g, 3.6 atm) E^∘ = 1.36 V (b) Fe^2 + (2M)

solutions - How to calculate the pH of a redox reaction between nickel(IV) oxide and silver? - Chemistry Stack Exchange
Calculate the equilibrium constant for the redox reaction at 25°C. Sr(s) + Mg^(2+) → Sr^(2+)(aq) + Mg(s), - Sarthaks eConnect | Largest Online Education Community

equilibrium - Calculate the cathode electrode potential in this redox reaction - Chemistry Stack Exchange


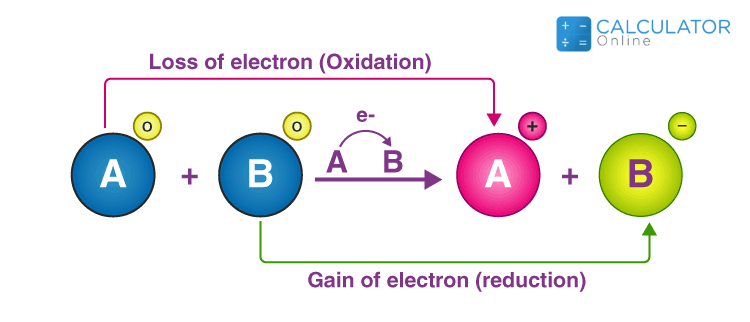

.png)