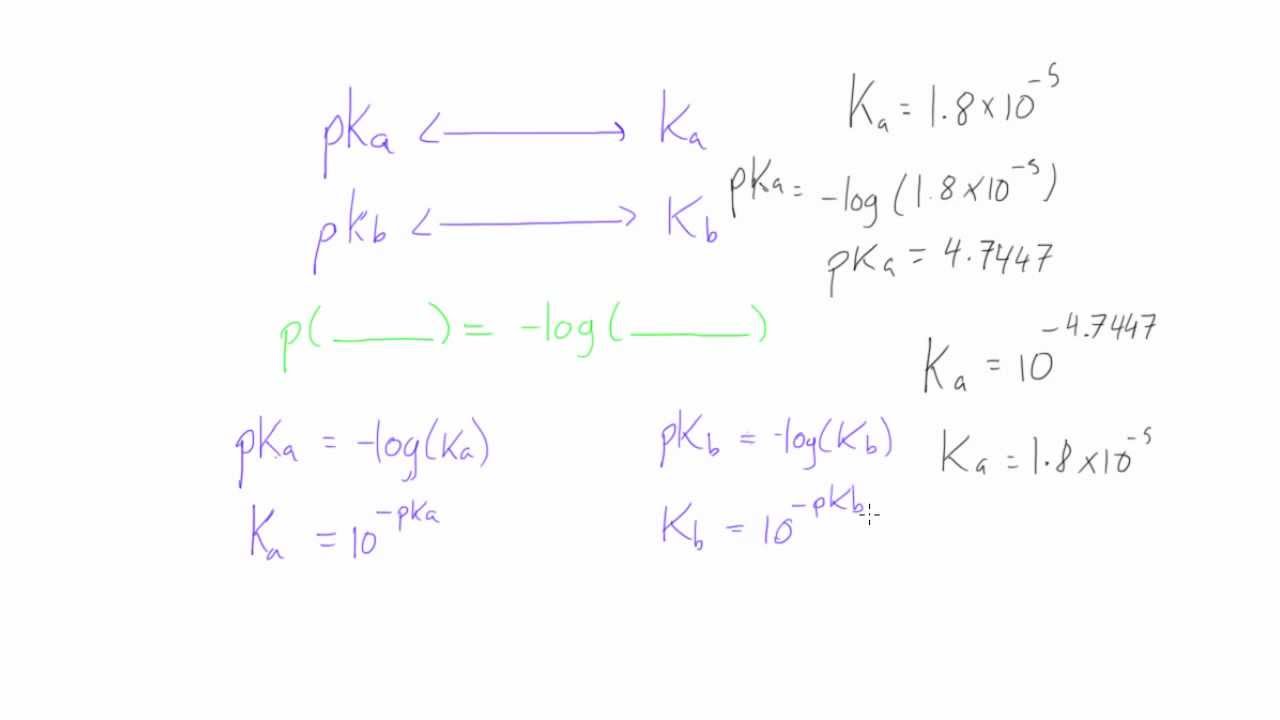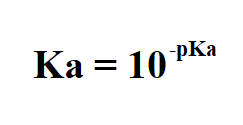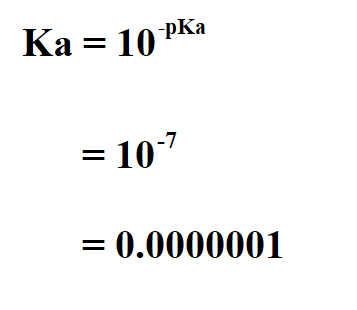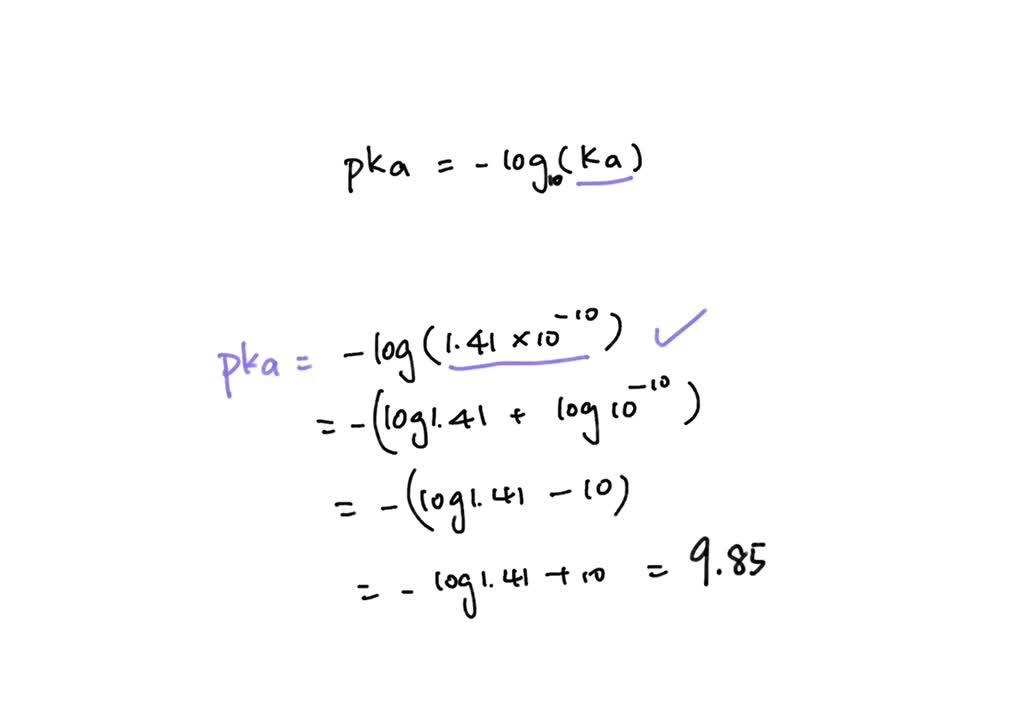
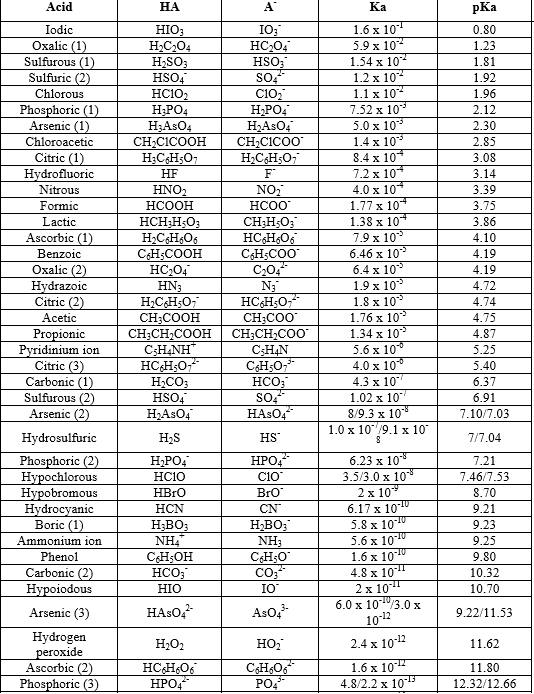
SOLVED: How would you calculate the pka or ka for CH2ClCH2COOH and CH2FCH2COOH if you only knew that this is a carboxylic acid? Do you write the conjugate acid and base to

SOLVED: Calculate Ka values for the following compounds. a) Tartaric acid ( pKa = 2.98) b) Phenol (pKa = 10.00)
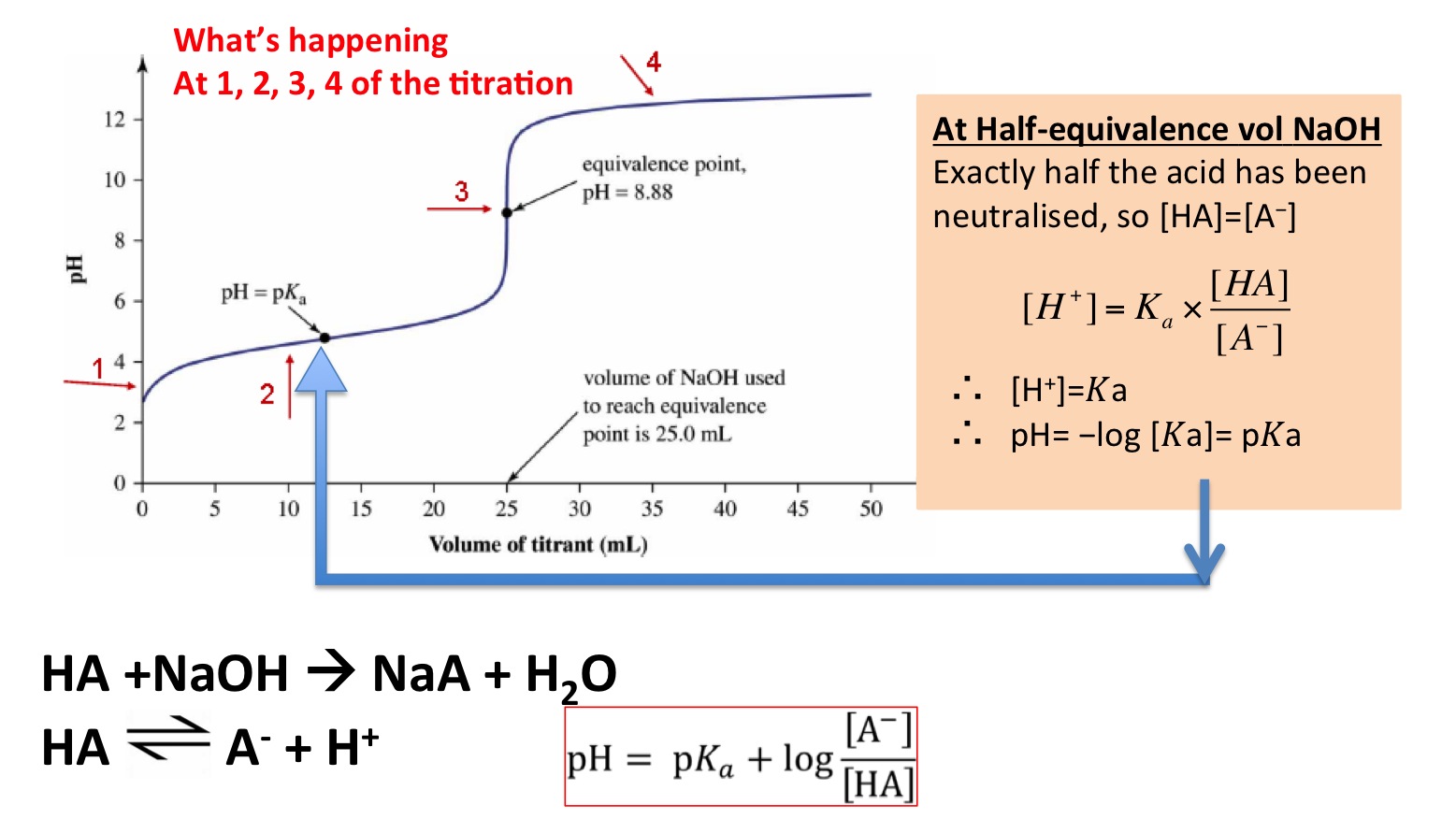

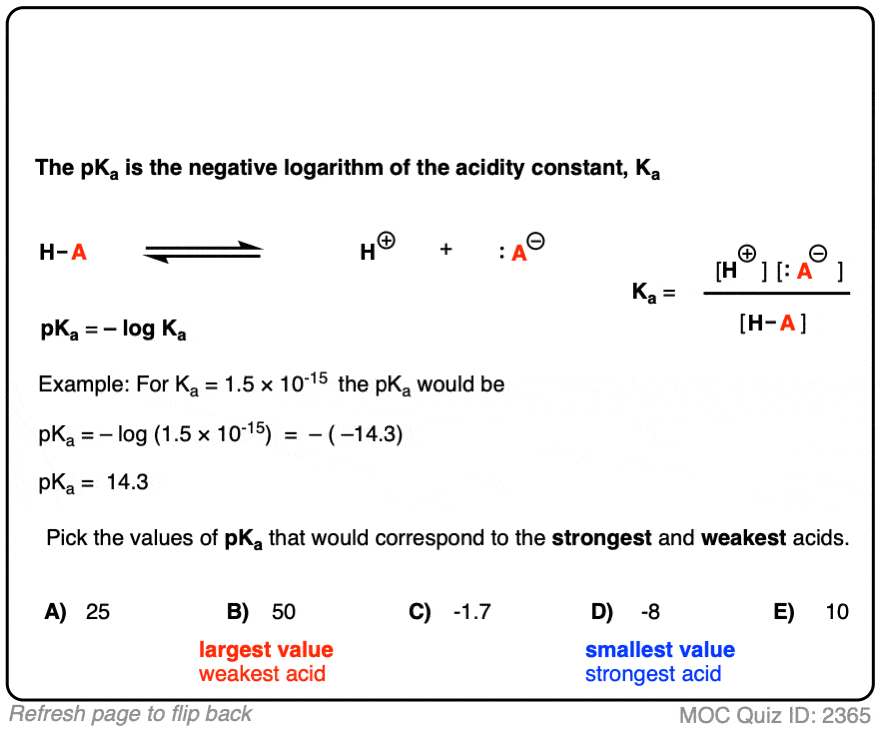
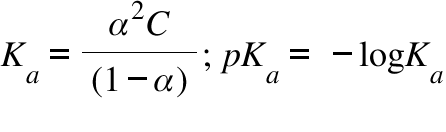

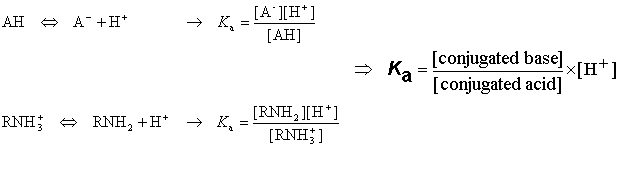
![Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube Biochemistry | Henderson-Hasselbalch Equation Proof [pH=pKa] - YouTube](https://i.ytimg.com/vi/2jpB30LsT8g/maxresdefault.jpg)