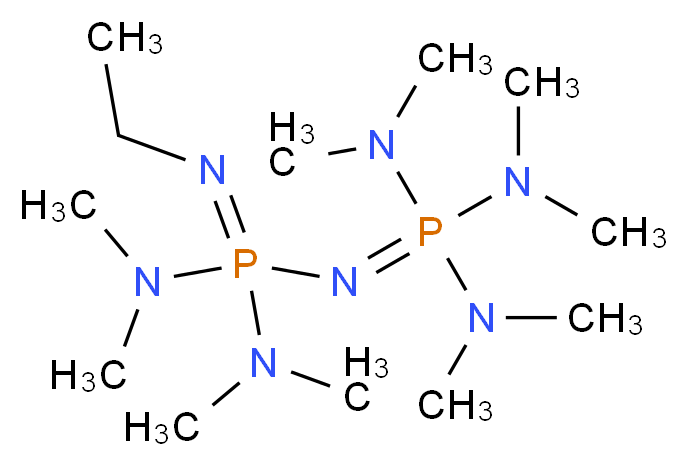
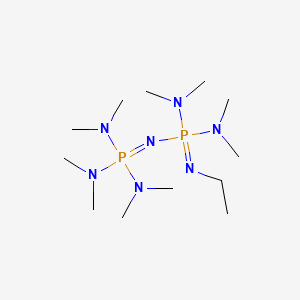
Tris(2,4,6-trimethoxyphenyl)phosphine - a Lewis base able to compete with phosphazene bases in catalysing oxa-Michael reactions | Organic Chemistry | ChemRxiv | Cambridge Open Engage

Organocatalytic Stereoselective Ring-Opening Polymerization of Lactide with Dimeric Phosphazene Bases | Journal of the American Chemical Society
![A Successful Application of Phosphazene Base P2‐tBu to [11C]ABP688 Radiosynthesis in Fully Automated Synthesis Module - Lee - 2020 - Bulletin of the Korean Chemical Society - Wiley Online Library A Successful Application of Phosphazene Base P2‐tBu to [11C]ABP688 Radiosynthesis in Fully Automated Synthesis Module - Lee - 2020 - Bulletin of the Korean Chemical Society - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/8d307dde-94b3-4266-9dd3-2fbd42ebce38/bkcs12070-fig-0002-m.jpg)