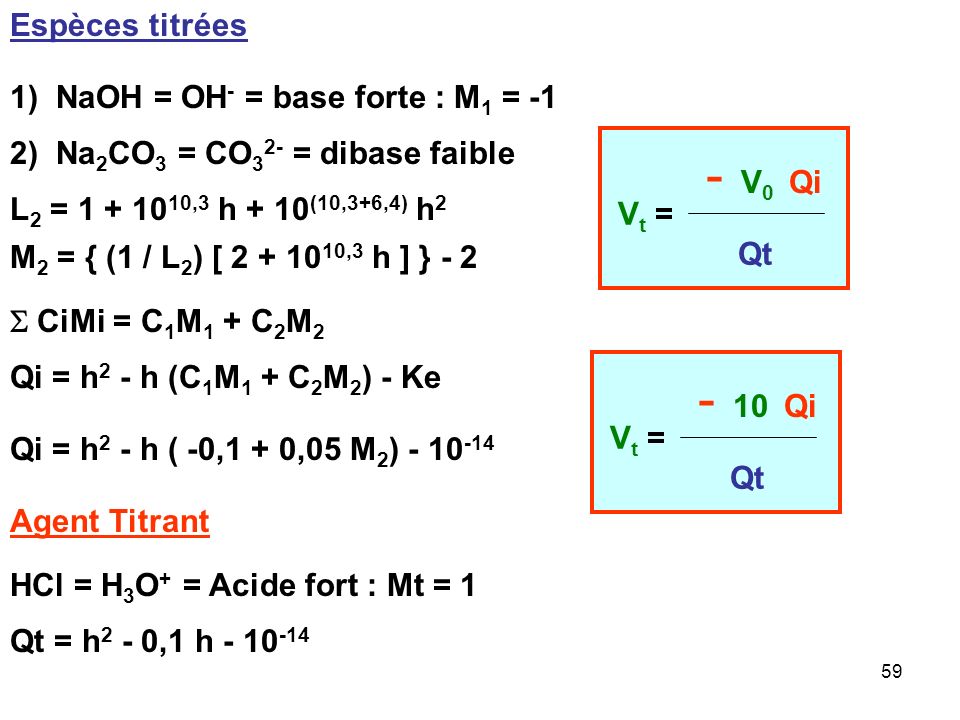
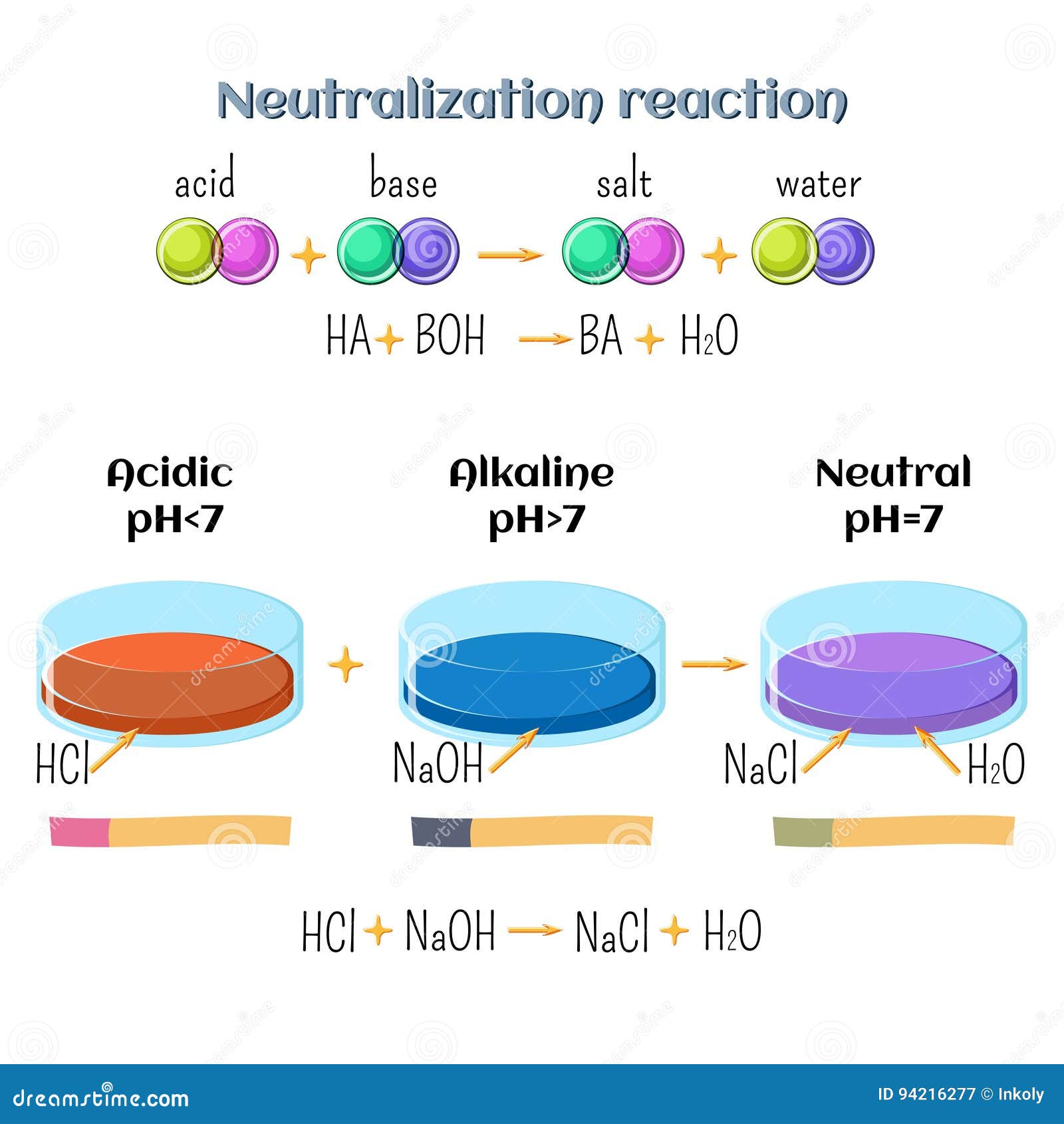
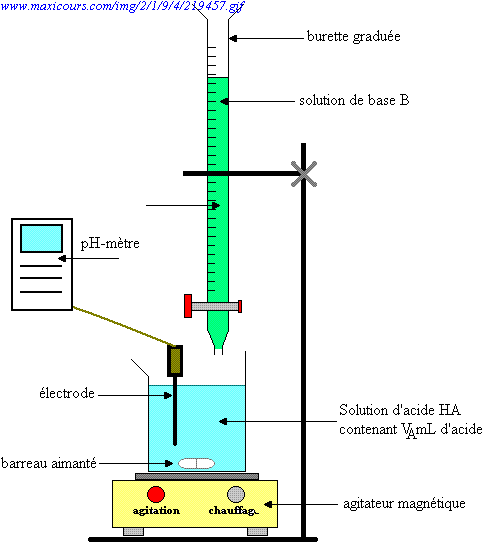
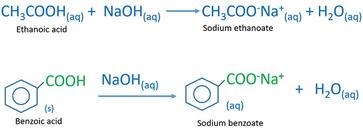
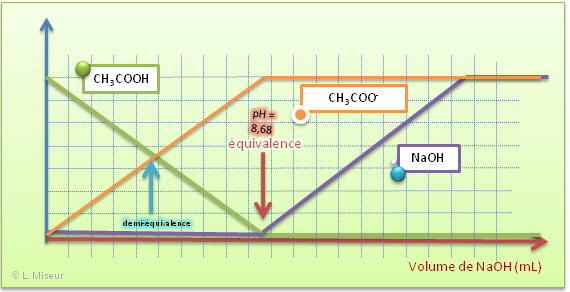
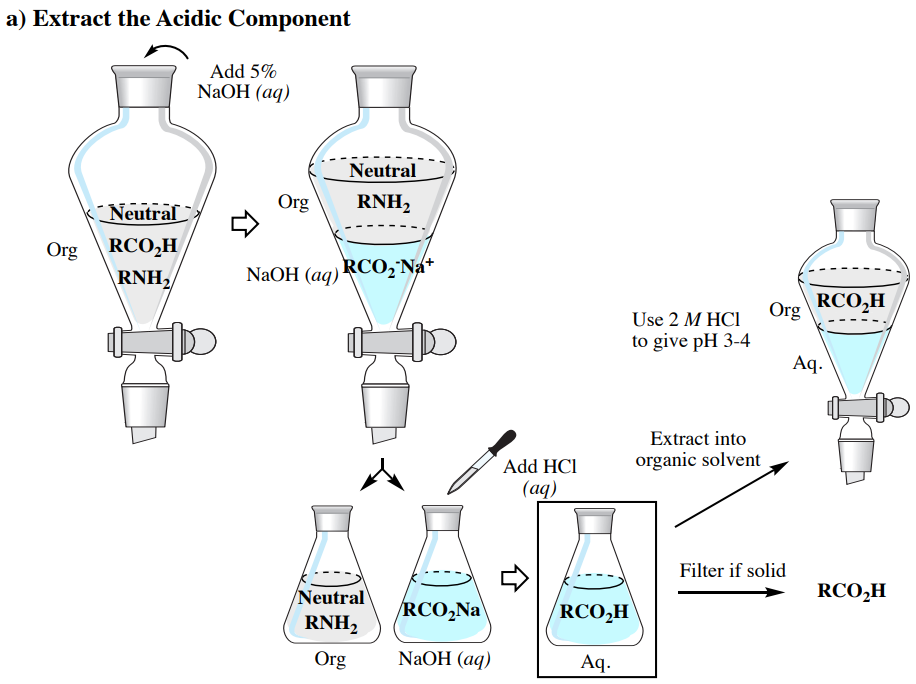
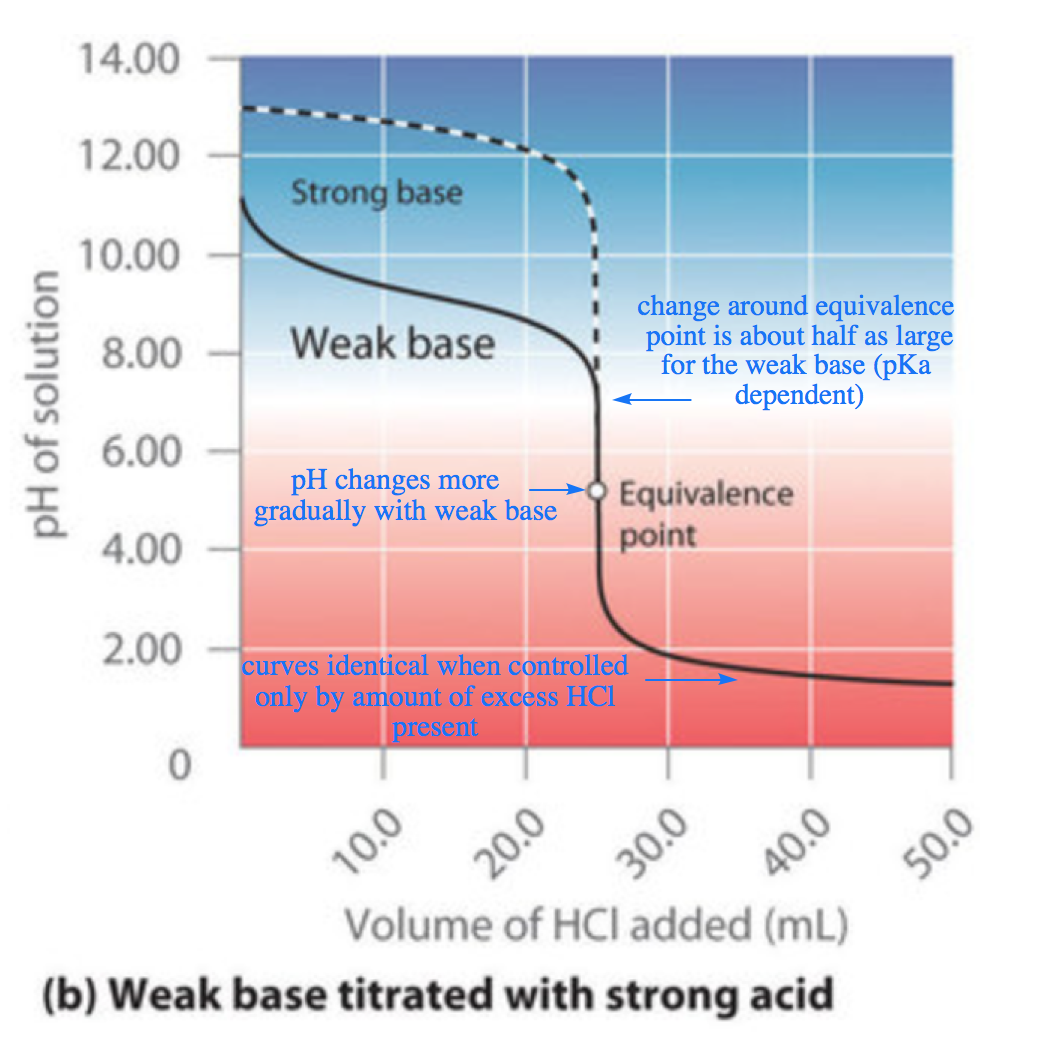
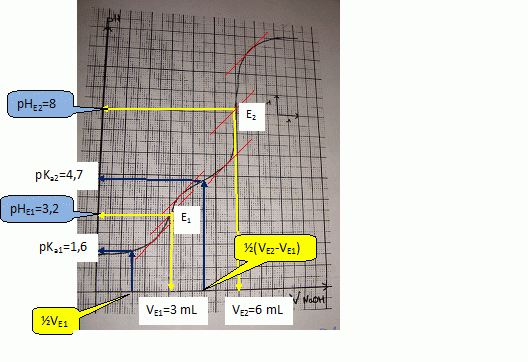
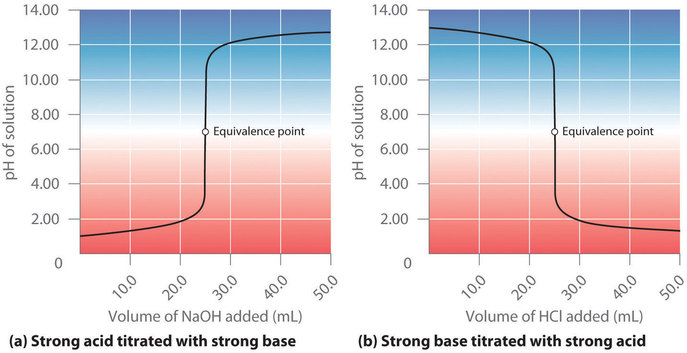
SOLVED: The balanced equation for the reaction between acetic acid and sodium hydroxide is shown below: HC2H3O2(aq) + NaOH(aq) à H2O(l) + Na C2H3O2(aq) Suppose that you perform a titration experiment to

Vidéo de question : Identifier le graphique montrant la variation du pH lors du mélange d'un acide avec une base | Nagwa