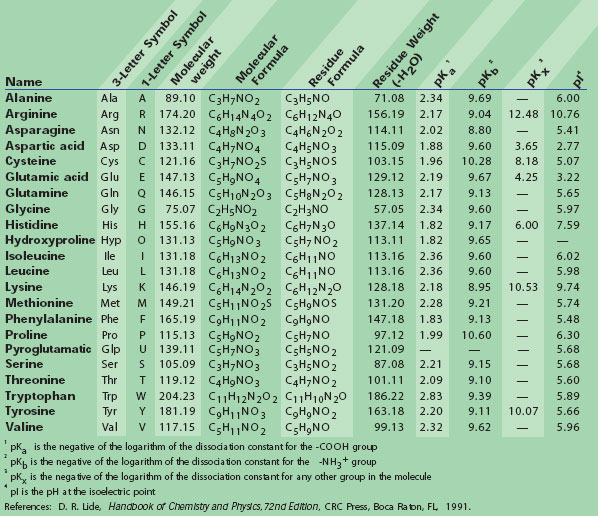
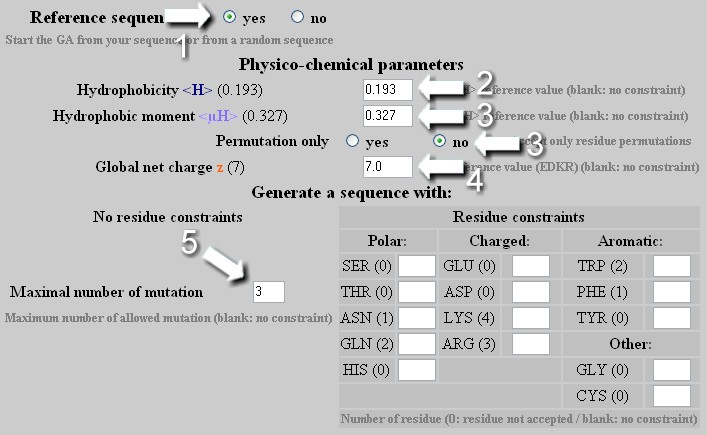
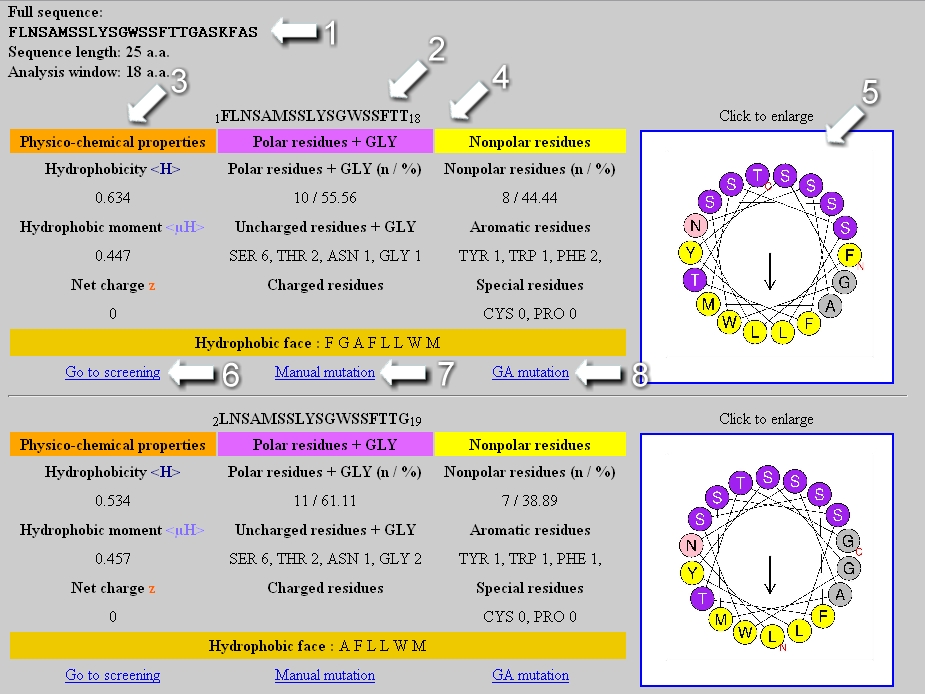
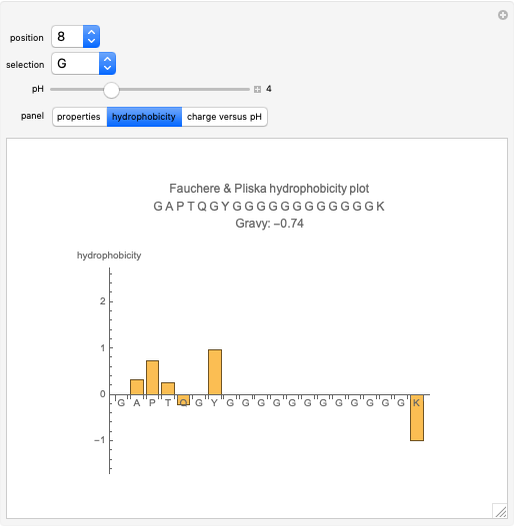
Methods of calculating protein hydrophobicity and their application in developing correlations to predict hydrophobic interaction chromatography retention - ScienceDirect

Solubility test of NSP and NSP r. (A) Peptide models computed by the... | Download Scientific Diagram

Peptide retention standards and hydrophobicity indexes in reversed-phase high-performance liquid chromatography of peptides. | Semantic Scholar

Analysis of hydrophobic and hydrophilic moments of short penetrating peptides for enhancing mitochondrial localization: prediction and validation - Pirisinu - 2019 - The FASEB Journal - Wiley Online Library

Analysis of hydrophobic and hydrophilic moments of short penetrating peptides for enhancing mitochondrial localization: prediction and validation - Pirisinu - 2019 - The FASEB Journal - Wiley Online Library

Probing the Outstanding Local Hydrophobicity Increases in Peptide Sequences Induced by Incorporation of Trifluoromethylated Amino Acids - Gadais - 2018 - ChemBioChem - Wiley Online Library
Protein Thermostability Is Owing to Their Preferences to Non-Polar Smaller Volume Amino Acids, Variations in Residual Physico-Chemical Properties and More Salt-Bridges | PLOS ONE
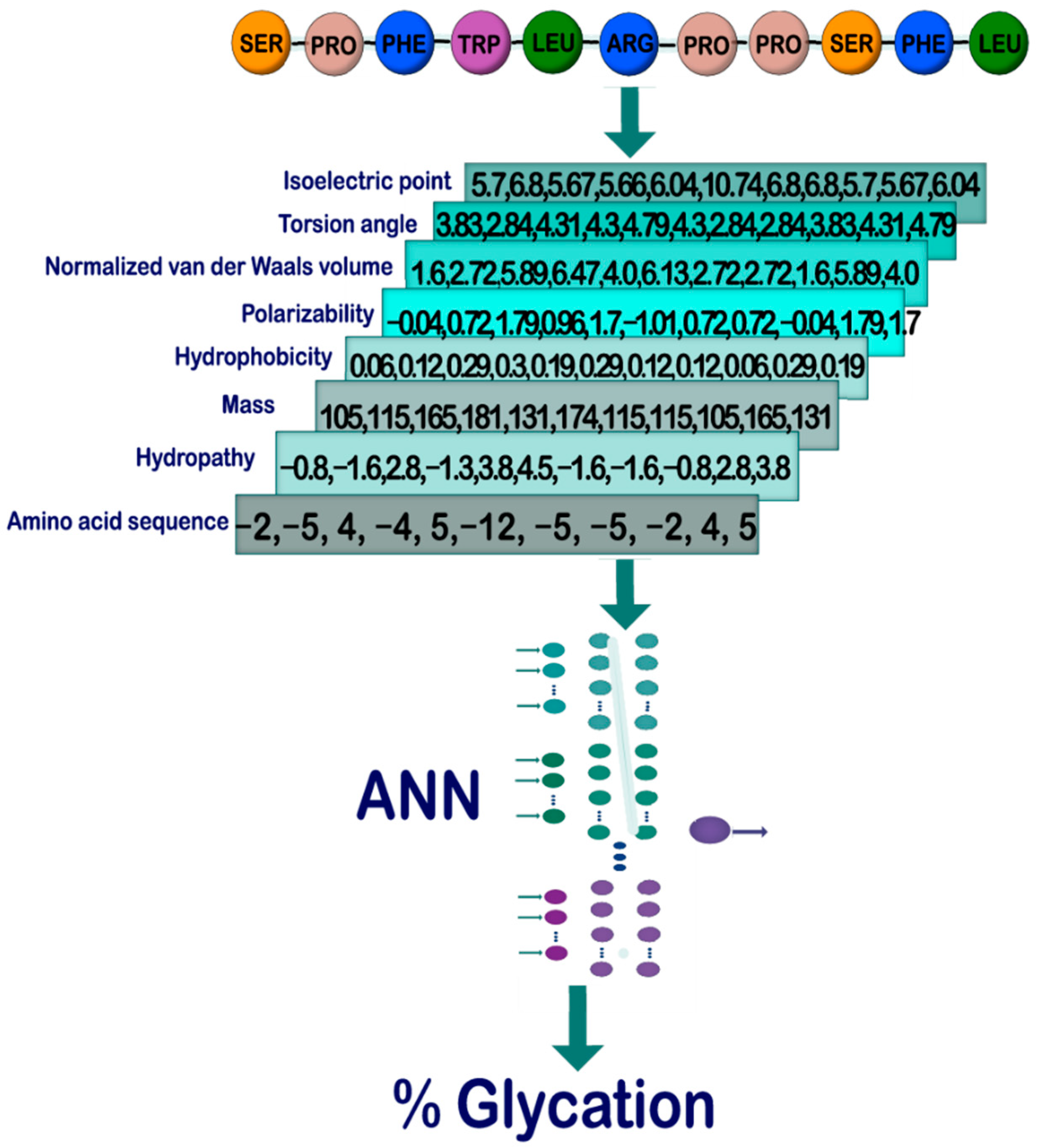
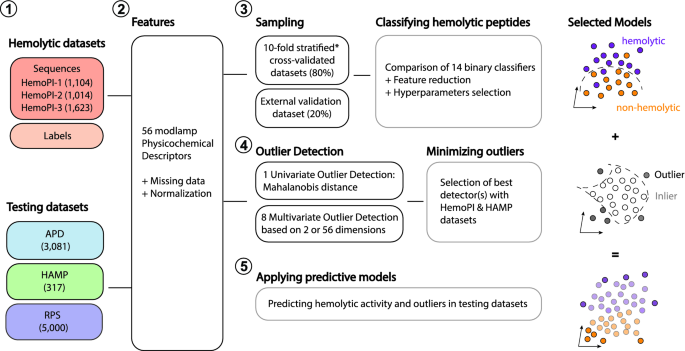
Sensors | Free Full-Text | On the Prediction of In Vitro Arginine Glycation of Short Peptides Using Artificial Neural Networks
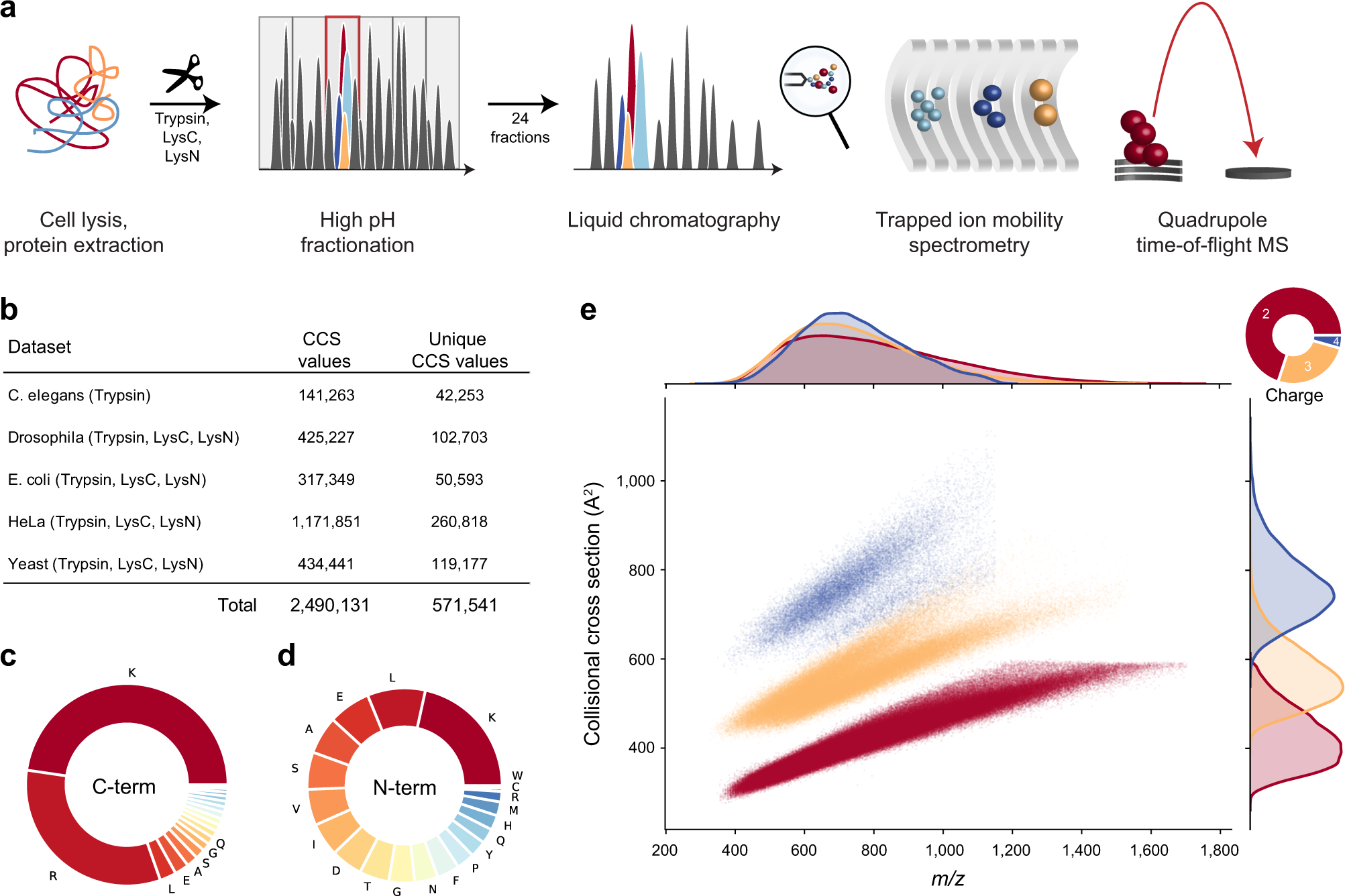
Deep learning the collisional cross sections of the peptide universe from a million experimental values | Nature Communications
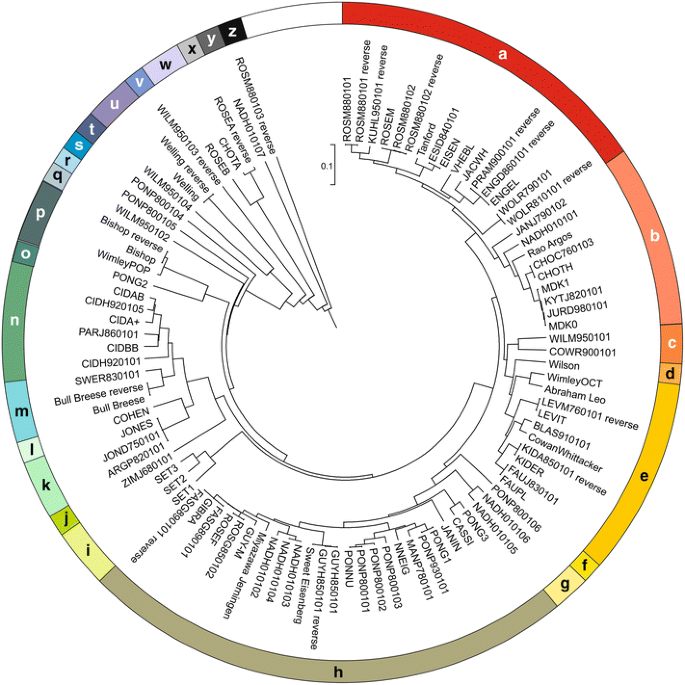
50 years of amino acid hydrophobicity scales: revisiting the capacity for peptide classification | Biological Research | Full Text

N-Capping Motifs Promote Interaction of Amphipathic Helical Peptides with Hydrophobic Surfaces and Drastically Alter Hydrophobicity Values of Individual Amino Acids | Analytical Chemistry

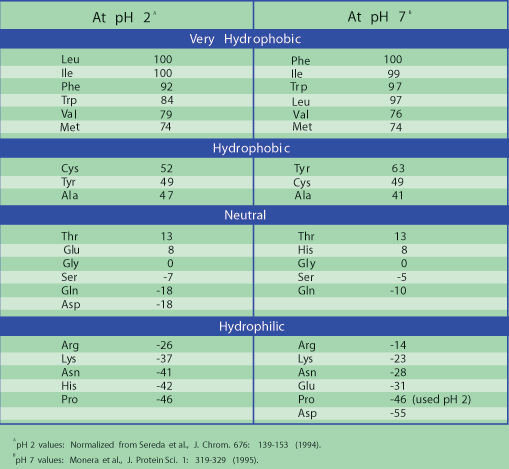




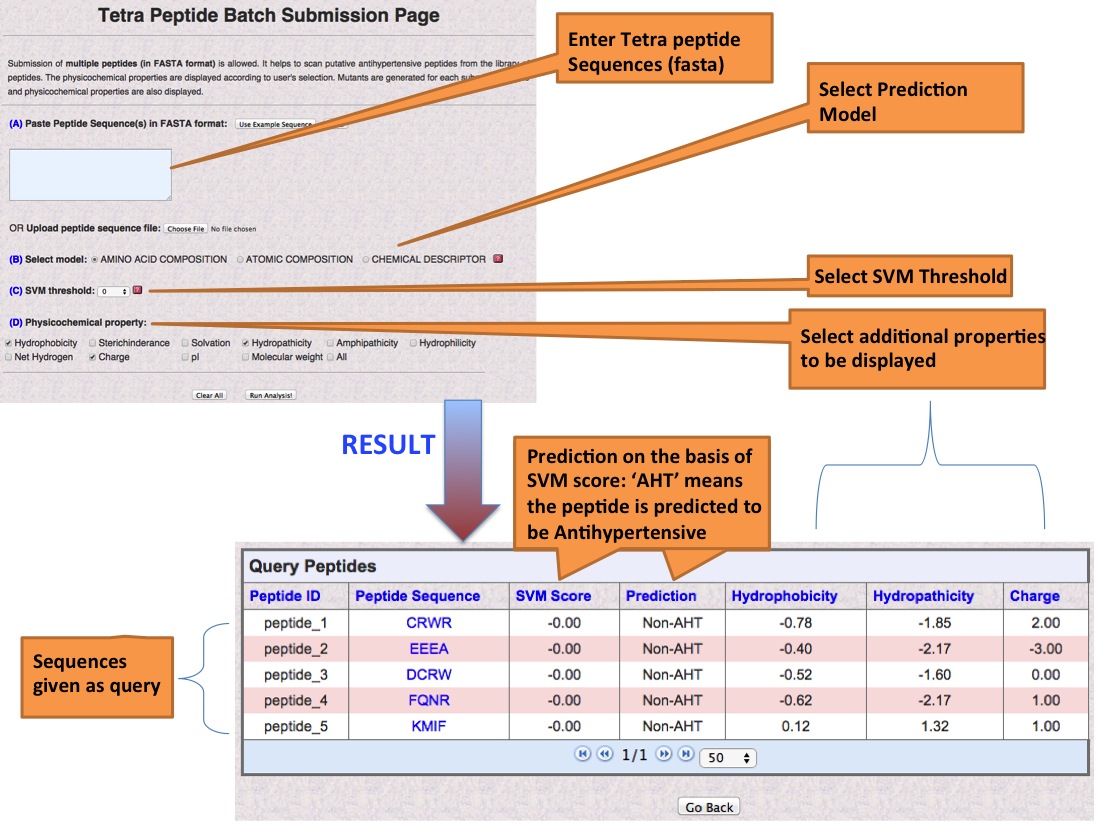
![PDF] Role of hydrophobicity in food peptide functionality and bioactivity | Semantic Scholar PDF] Role of hydrophobicity in food peptide functionality and bioactivity | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/107c6e0004c5d8ceebd70e09539a91b7089fb82d/3-Table2-1.png)