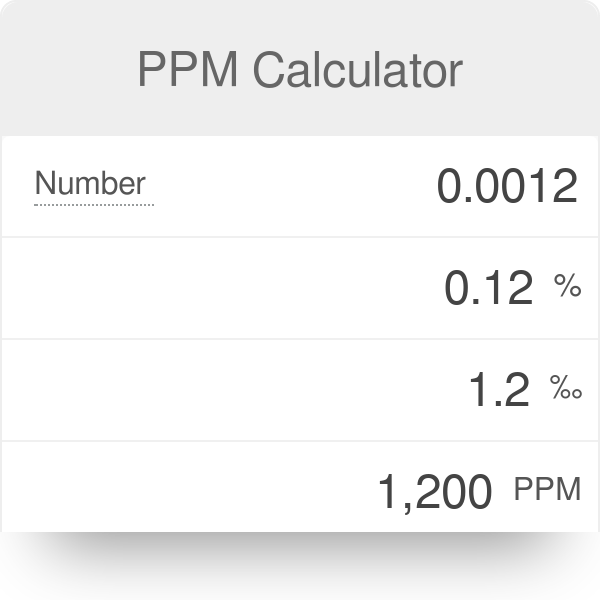
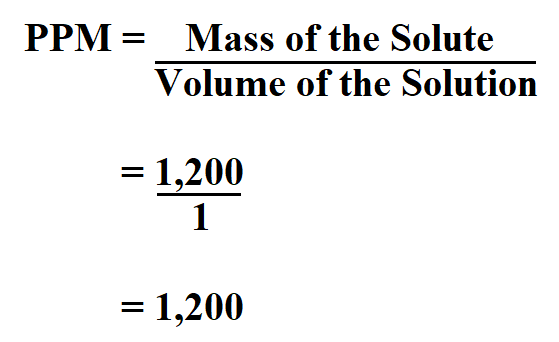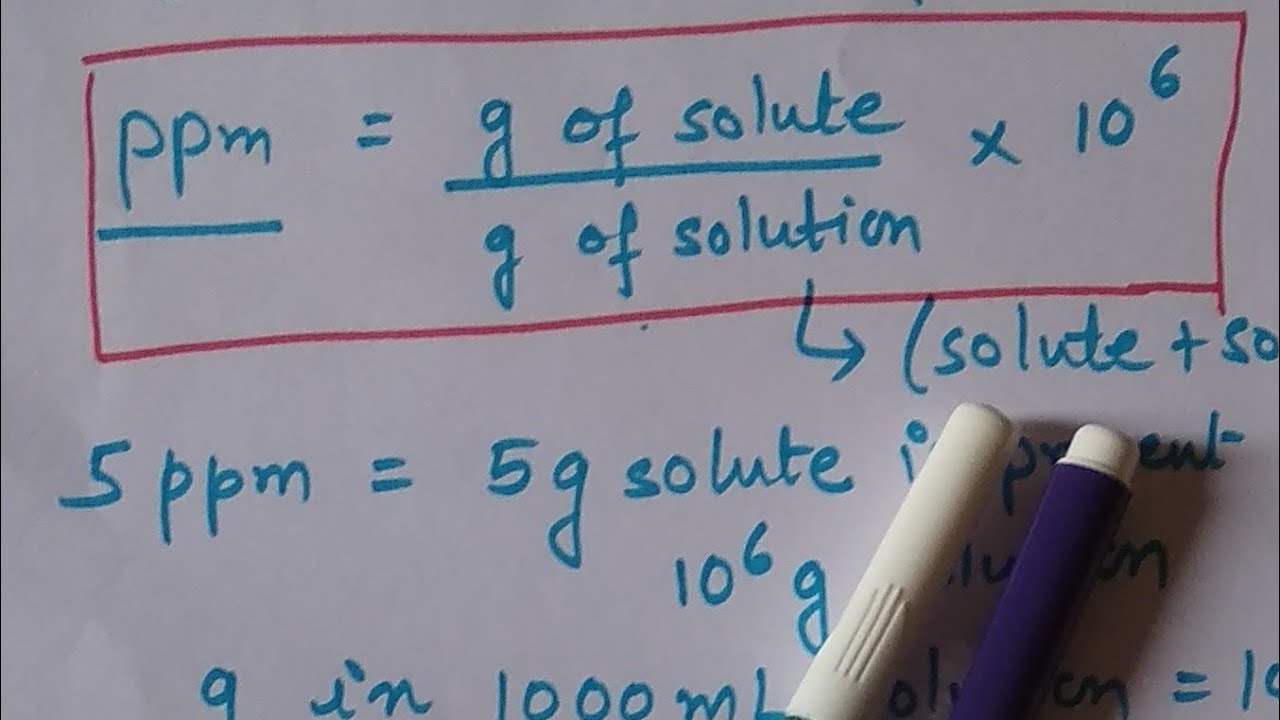A water sample is found to contain 96 ppm of SO4^(2-) and 122 ppm of HCO+3^(ɵ) with Ca^(2) ion as the only cation. (a). Calculate the ppm of Ca^(2+) in water. (b).

Calculate the ppm of mercury in water in given sample contain `30mg` of `Hg` in `500ml` of solution - YouTube

SOLVED: Calculate the concentration of Fe (ppm) on a diluted tablet solution given that the intercept of an Absorbance vs. Concentration (ppm) plot has a slope of 0.0083 ppm-' with and an