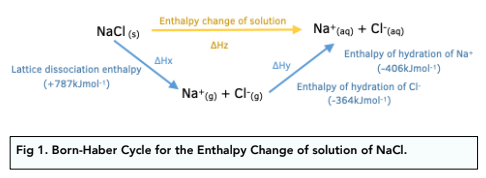
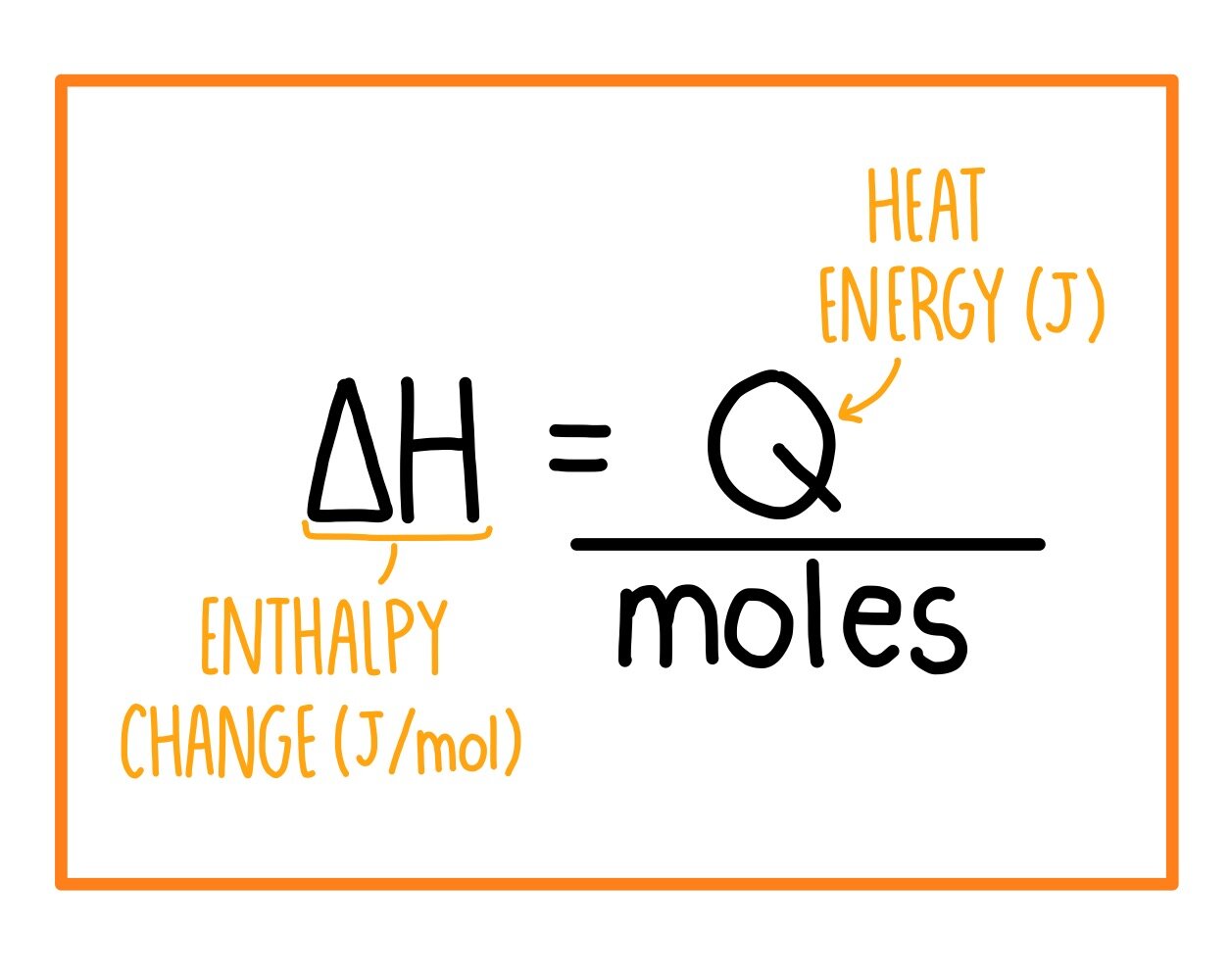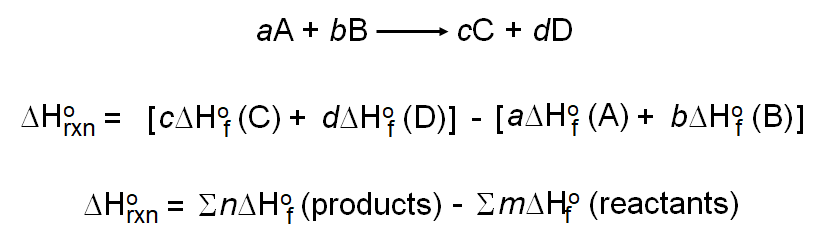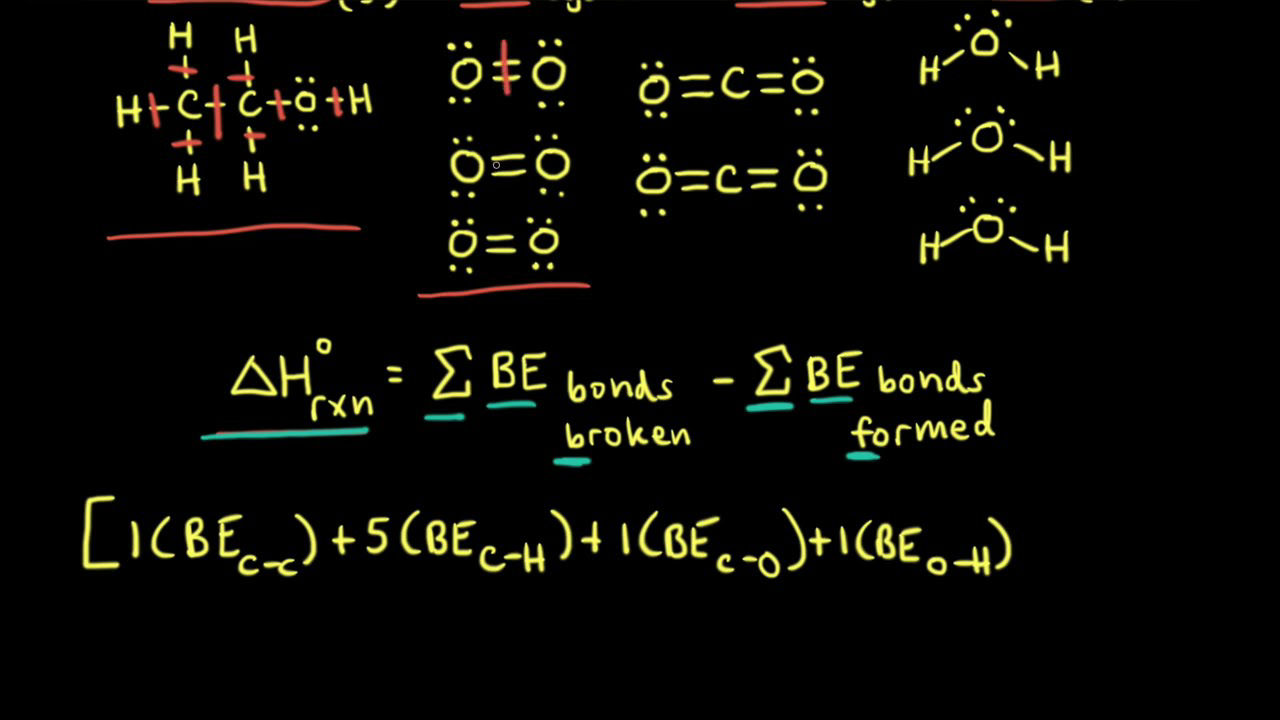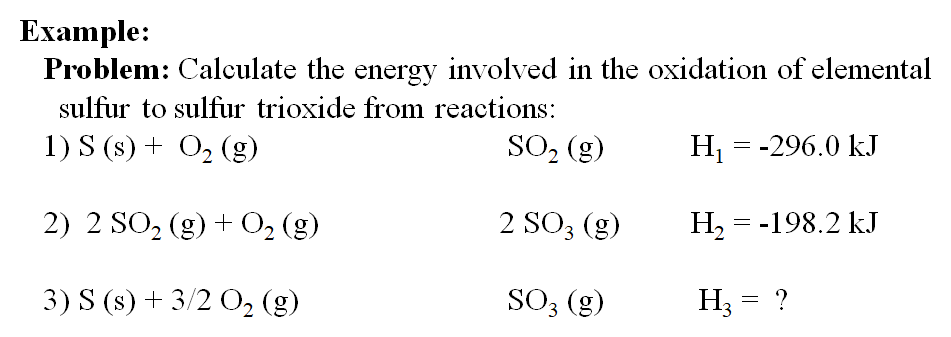thermodynamics - Does the enthalpy of solution formula (LE + Hyd) change depending on the question? - Chemistry Stack Exchange

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry

Calculate the enthalpy of hydrogenation : C(2)H(5)(g) + H(2)(g) to C(2)H(6)(g) Given that the bond enthalpies of H - H , C = C, C- C and C - H bonds are
By the end of today's lesson you should · know what enthalpy is · · · understand the difference between a molar enthalpy v
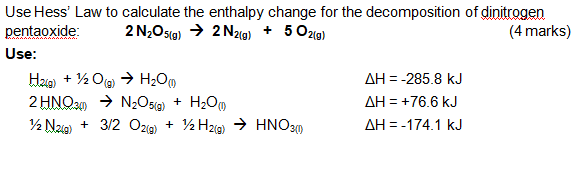
Use Hess' Law to calculate the enthalpy change for the decomposition of dinitrogen pentaoxide? | Socratic