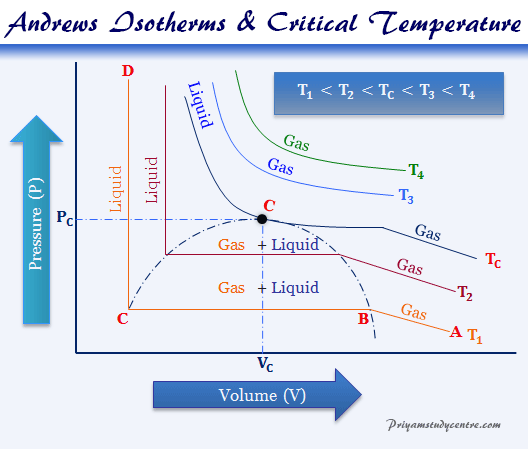
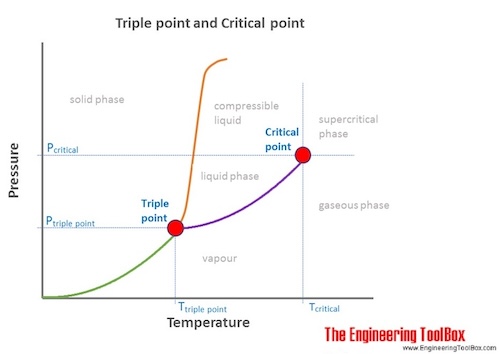
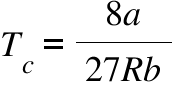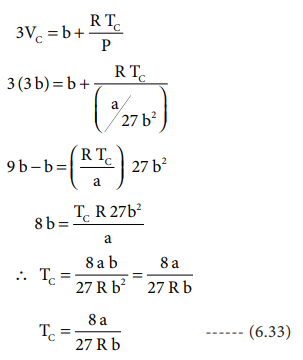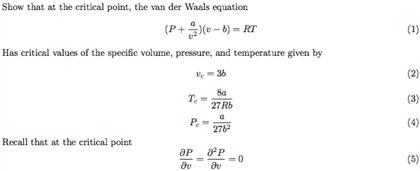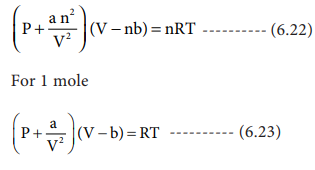Chemical & Process Technology: Removal of Specific Heat ratio (k) in the Mach No. & Critical Pressure Calculation

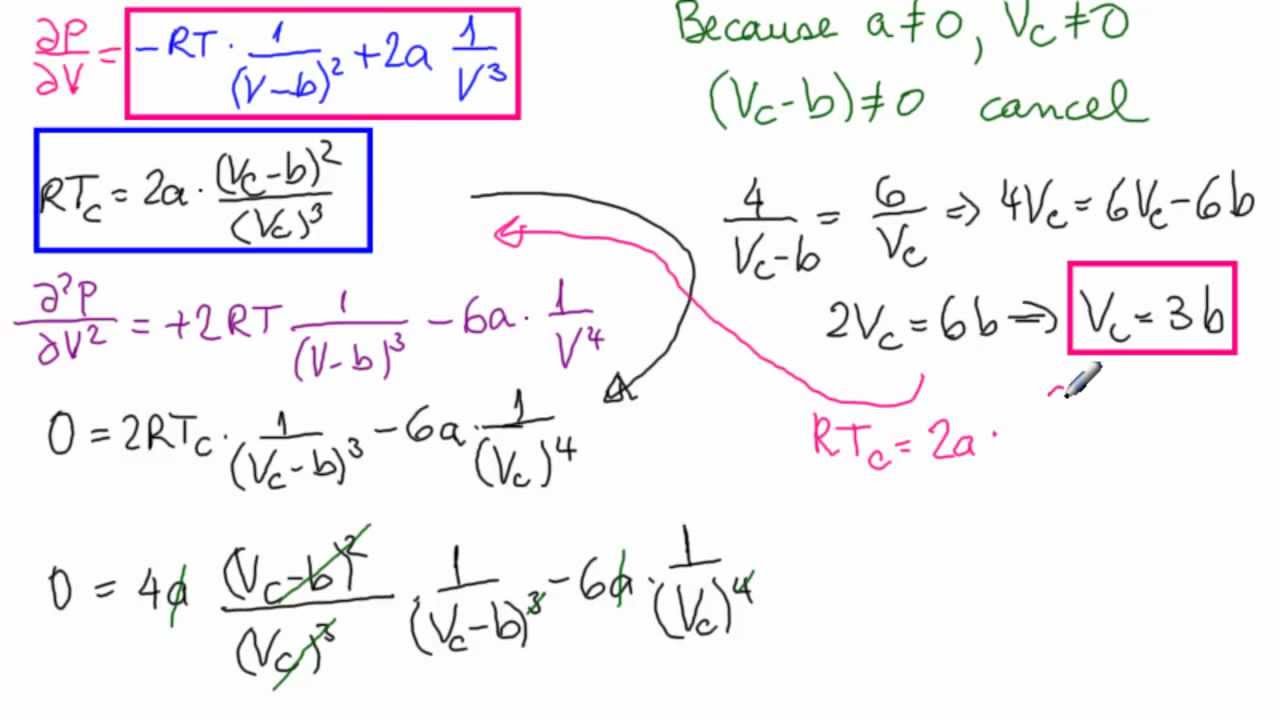
The values of the van der Waals constants for a gas a = 4.10 dm^(6) "bar" mol^(-2) and b = 0.035 dm^(3) mol^(-1). Calculate the values of the critical temperature and critical
Chemical & Process Technology: Removal of Specific Heat ratio (k) in the Mach No. & Critical Pressure Calculation

Methane Gas Volume Expansion Ratios and Ideal Gas Deviation Factors for the Deep-Water Bering Sea Basins: Peng-Robinson Equation of State

Calculate critical temperature and critical pressure of oxygen if the values of the van der - YouTube

For a real gas critical pressure is 75 atm and van der Waal's constant 'b' is 40 millilitres per - YouTube
The critical temp. and pressure for NO gas at 177 k and 64.5 atm respectively. Calculate van der Waals constant 'a' & 'b' for the gas. - Sarthaks eConnect | Largest Online Education Community