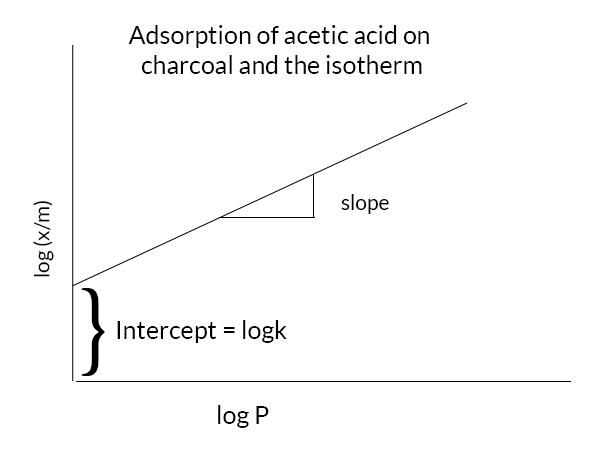
4. Adsorption Equilibria Examples - Example 1 An adsorption study is set up in laboratory by adding a known amount of activated carbon to six flasks | Course Hero
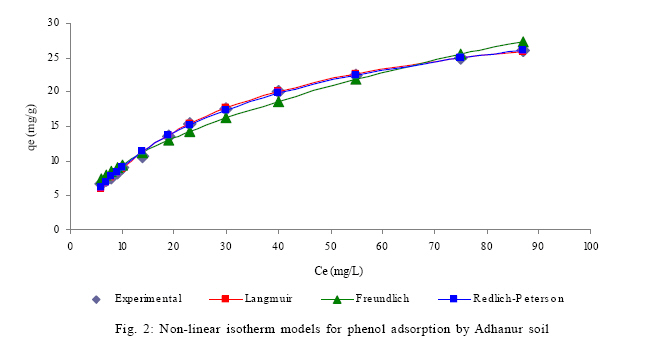
How to Calculate Adsorption Isotherms of Particles Using Two-Parameter Monolayer Adsorption Models and Equations Indonesian Jour

Optimization and Experimental Design of the Pb2+ Adsorption Process on a Nano-Fe3O4-Based Adsorbent Using the Response Surface Methodology | ACS Omega

Adsorption Equilibrium and Kinetics of the Removal of Ammoniacal Nitrogen by Zeolite X/Activated Carbon Composite Synthesized from Elutrilithe

Linear and nonlinear kinetic and isotherm adsorption models for arsenic removal by manganese ferrite nanoparticles | SpringerLink
How to Calculate Adsorption Isotherms of Particles Using Two-Parameter Monolayer Adsorption Models and Equations Indonesian Jour
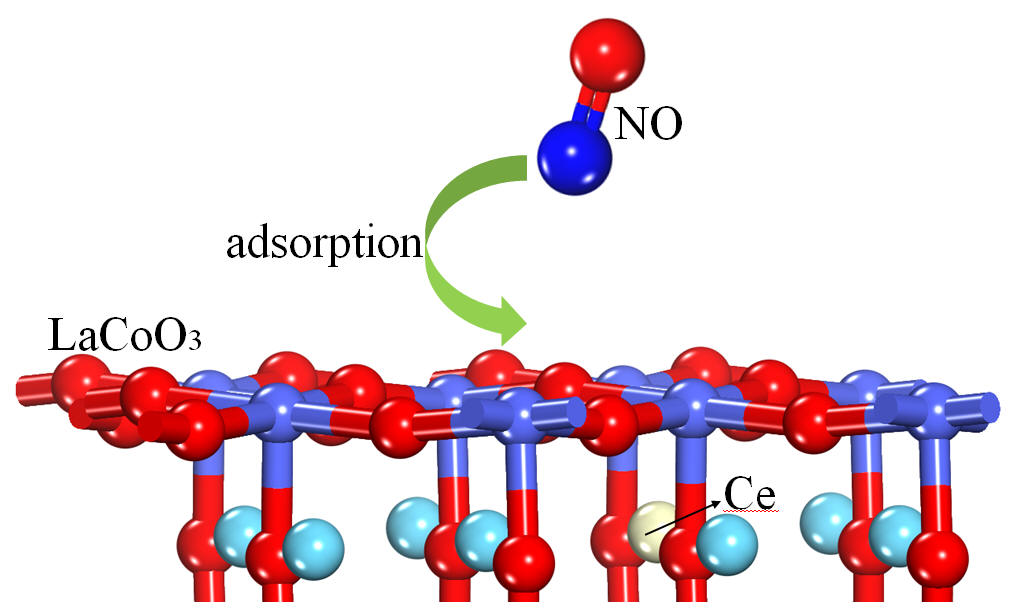
Materials | Free Full-Text | DFT Analysis of NO Adsorption on the Undoped and Ce-Doped LaCoO3 (011) Surface

Figure 6 from Synthesis of poly(ortho-phenylenediamine) fluffy microspheres and application for the removal of Cr(VI) | Semantic Scholar
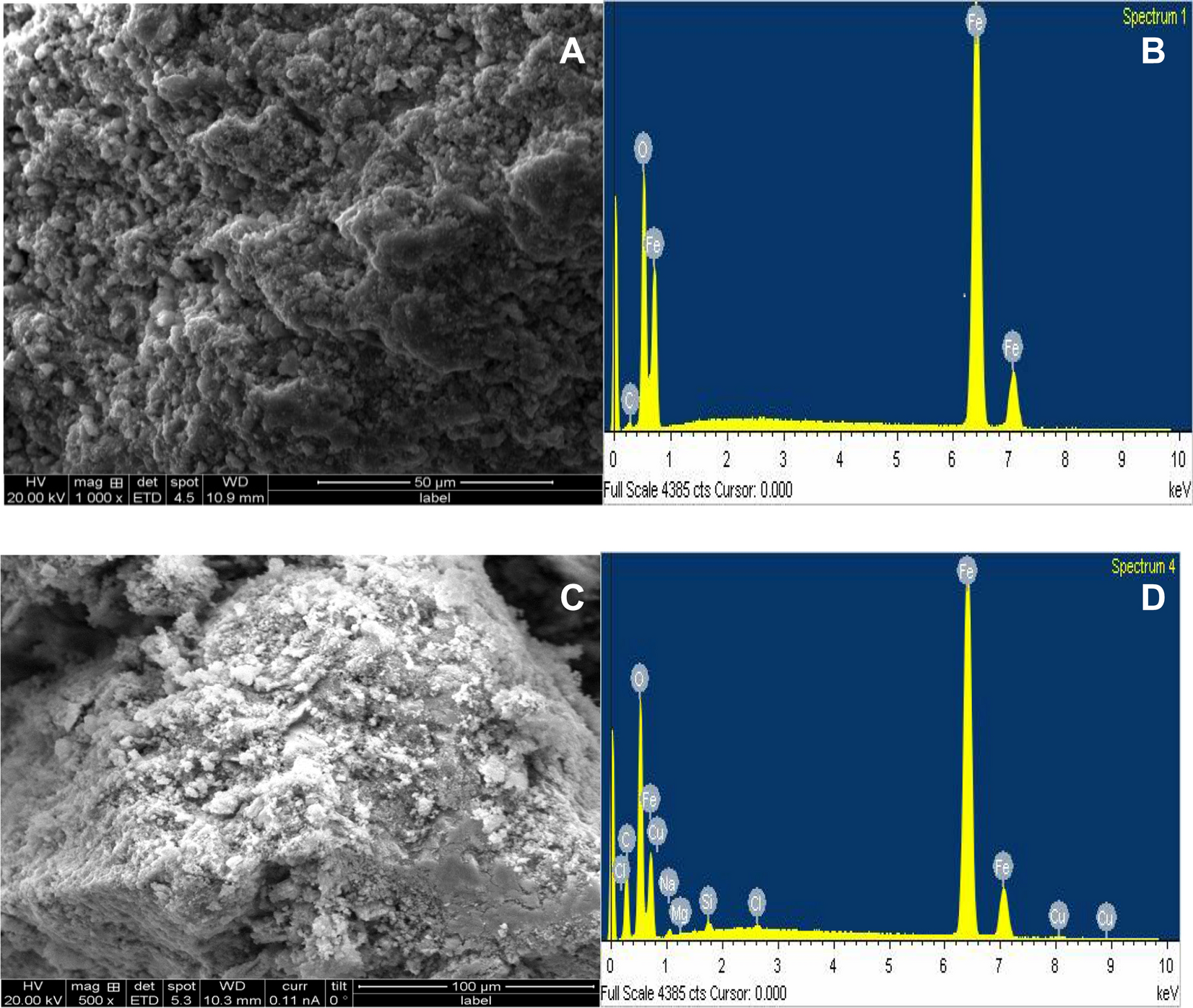






%20of%20langmuir%20isotherm.jpg?ezimgfmt=rs:363x257/rscb1/ngcb1/notWebP)