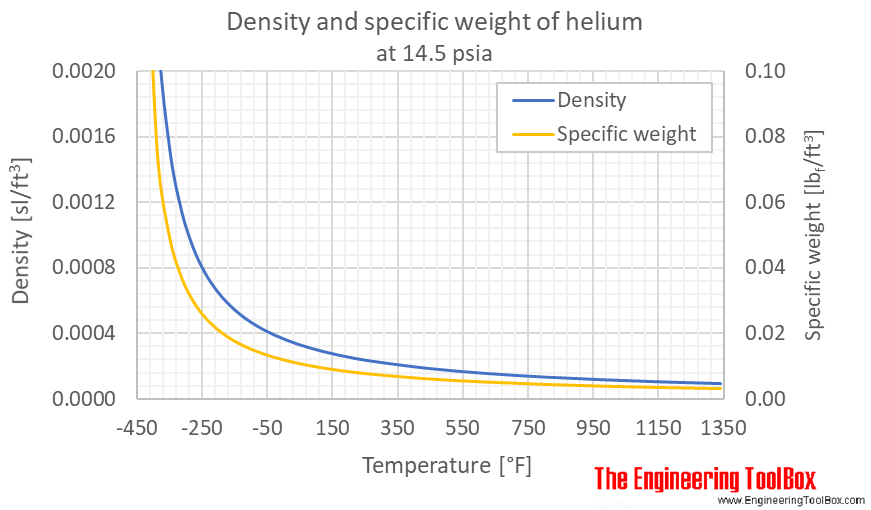
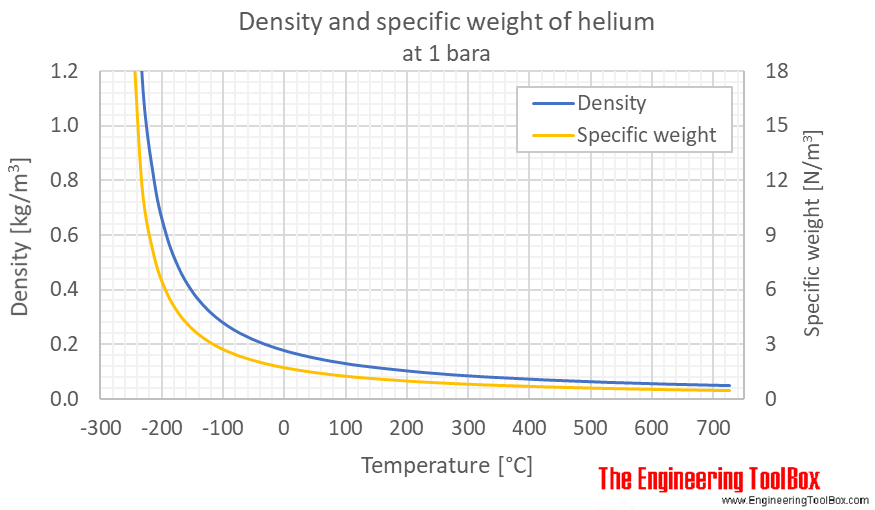
A large balloon of mass 226 kg is filled with helium gas until its volume is 325 m3. Assume the dens - YouTube

Ideal Vacuum | EDWARDS ELD500 Helium Leak Detector w/ internal Diaphragm DRY Pump 100-120 VAC 60 Hz PN D13520906

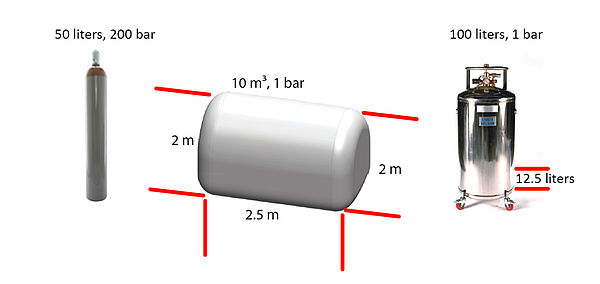
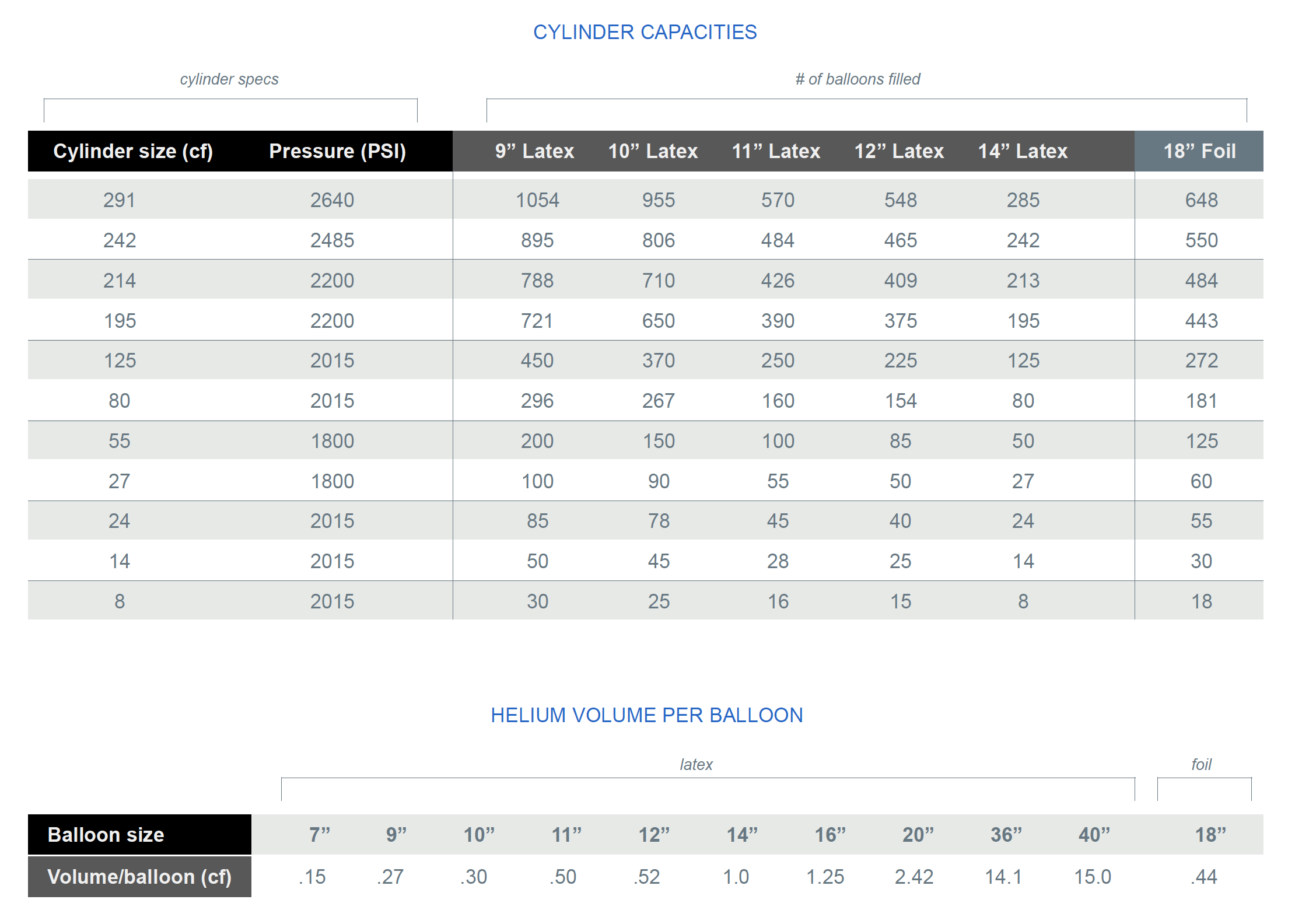
The Very Best Balloon Blog: Helium - A little bit of what you need to know as a balloon professional Part 2.
The Very Best Balloon Blog: Helium - A little bit of what you need to know as a balloon professional Part 2.

A balloon initially contains 65 m^3 of helium gas at atmospheric conditions of 100 kPa and 22 degrees C. The balloon is connected by a valve to a large reservoir that supplies