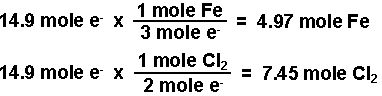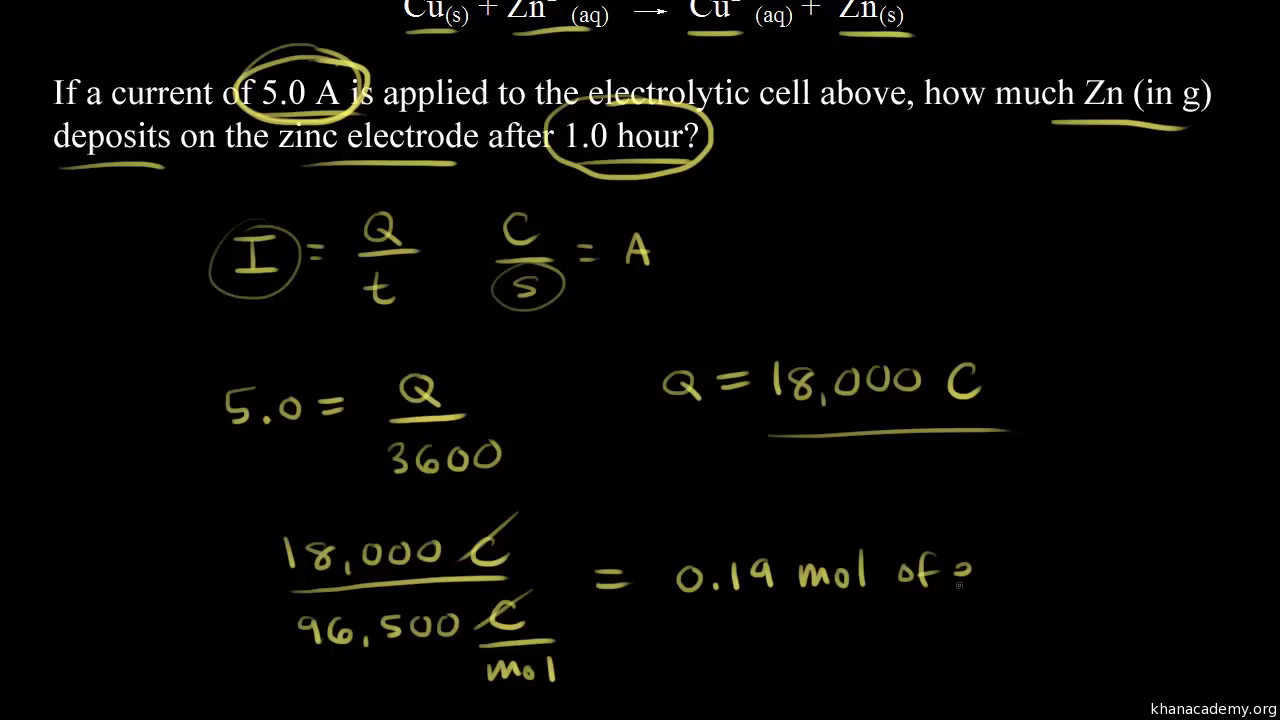Question Video: The Ionic Equation for the Electrolysis of the Molten Salt of Barium at the Negative Electrode | Nagwa

SOLVED: Electrolysis of aqueous solution of 0.1 M CuBrz was carried out using inert electrode at 25 Determine the products of electrolysis at anode and cathode: Justify. Calculate the mass of product
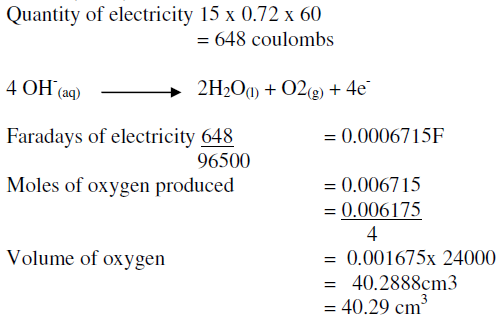
During the electrolysis, a current of 0.72A was passed through the electrolyte for 15 munites.Calculate the volume of gas produced at the anode.(1 Faraday = 96...