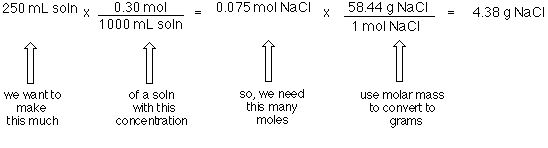If the concentration is expressed in moles per liter, the unit of the rate constant for a first- - YouTube

Concentration. Concentration Particles per volume Can be in grams per litre but chemists usually express concentration in moles per litre This are related. - ppt download

Calculate the concentration of nitric acid in moles per litre in a sample which has density 1.41g/mL - YouTube

Molarity 2. Molarity (M) this is the most common expression of concentration M = molarity = moles of solute = mol liters of solution L Units are. - ppt download

What is the approximate pH of a solution if the concentration of hydrogen ions is 5.0x10^-4 moles per - Brainly.com

PPT - MOLARITY A measurement of the concentration of a solution Molarity (M) is equal to the moles of solute (n) per liter of PowerPoint Presentation - ID:442848

SOLVED: 32 Using the following equation, determine the average concentration (moles per liter) of Ca2+ ions present in your water: Record the concentration in Data Table 2- mol Ca2+ mol EDTA =

Calculate the concentration of nitric acid in moles per litre in a sample which has a density of 1.41g mL^-1 and the mass per cent of nitric acid in it being 69
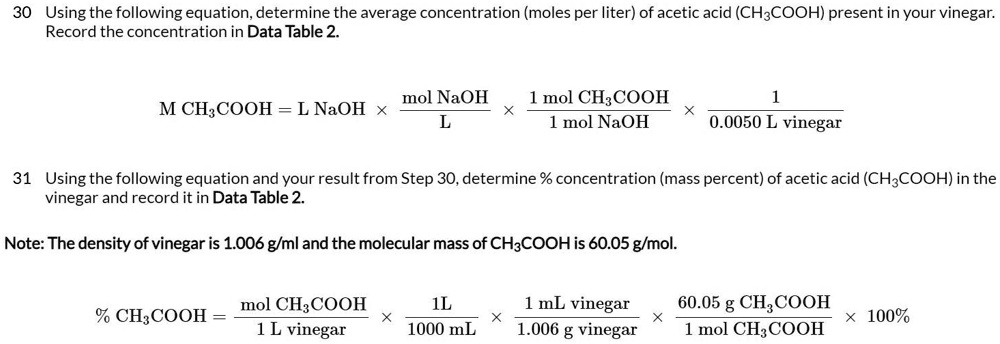
SOLVED: 30 Using the following equation; determine the average concentration (moles per liter) of acetic acid (CHzCOOH) present in your vinegar: Record the concentration in Data Table 2 mol NaOH mol CH:COOH

Calculate the concentration of nitric acid in moles per litre in a sample which has a density 1.41 g mL^-1 and the mass per cent of nitric acid in it being 69% .