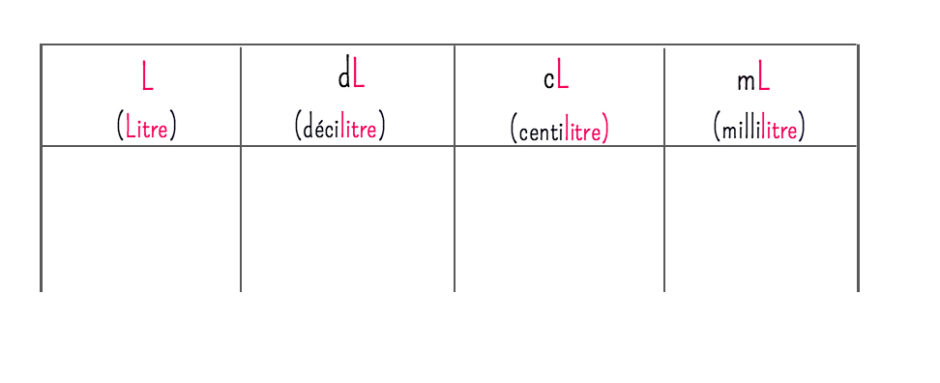
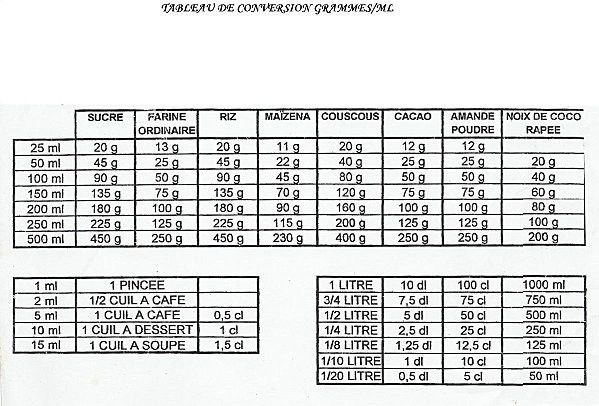
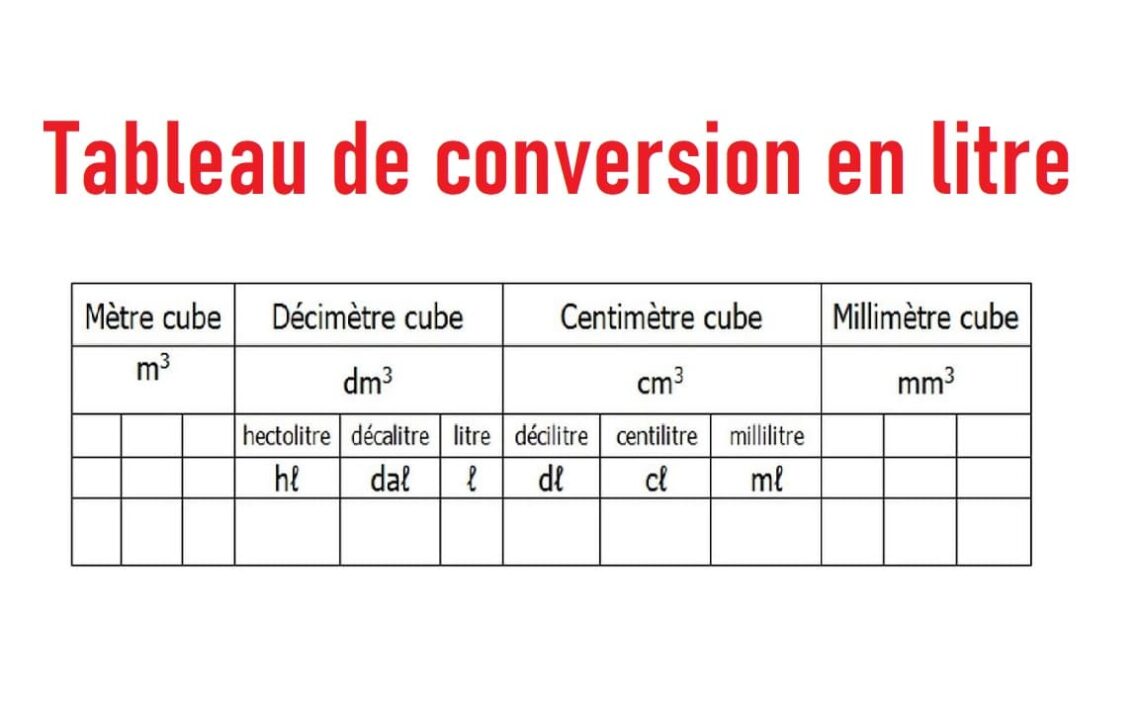
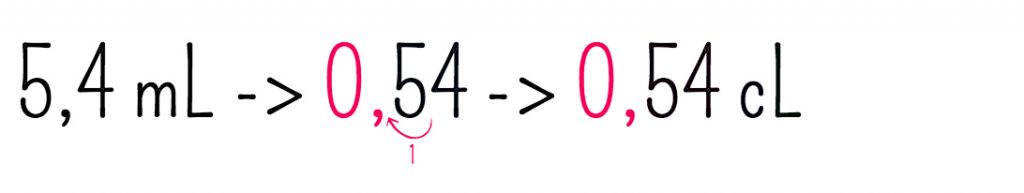SOLVED: You added 0.05 mL of 0.10 M AgNO, t0 4.0 mL of 2.0 M NaCl, and formed saturated solution of AgCI (s): AgCI (s) (2q)+Cl- (aq) K= Xsp Using this saturated

Expanding the Polymethine Paradigm: Evidence for the Contribution of a Bis-Dipolar Electronic Structure | The Journal of Physical Chemistry A
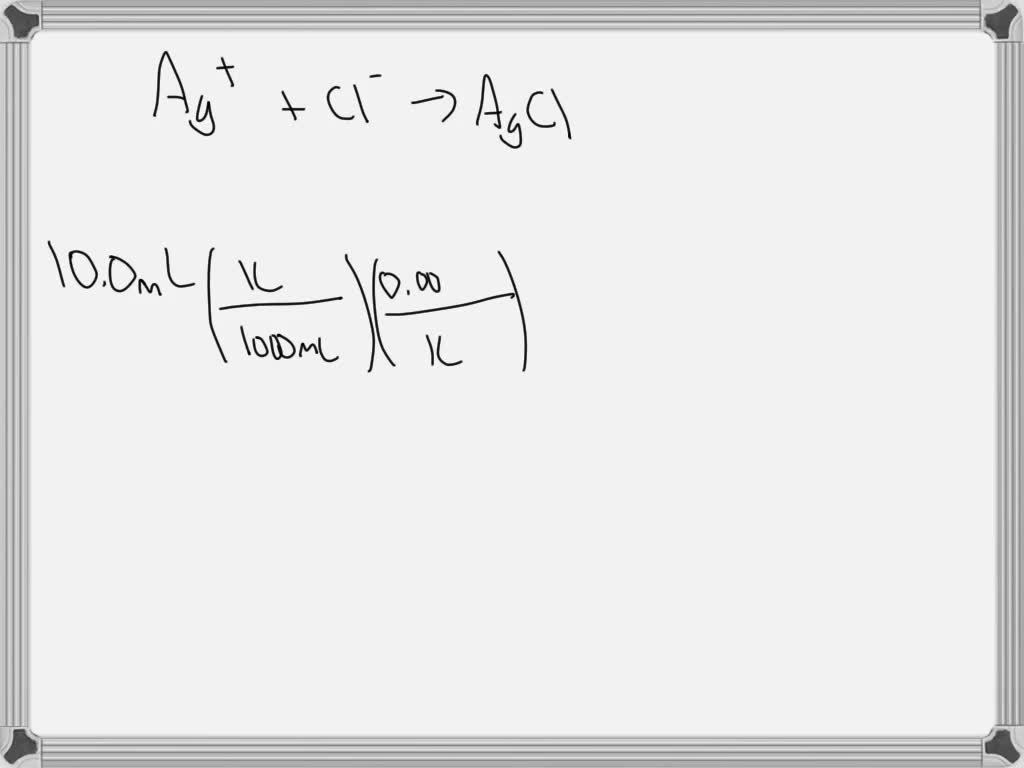
SOLVED: A volume of 10.0 mL of a 0.00350 M solution of Cl– ions are reacted with a 0.500 M solution of AgNO3. What is the maximum mass of AgCl that precipitates?