![Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download](https://images.slideplayer.com/21/6241563/slides/slide_11.jpg)
Acid-Base balance Prof. Jan Hanacek. pH and Hydrogen ion concentration pH [H+] nanomol/l ppt download

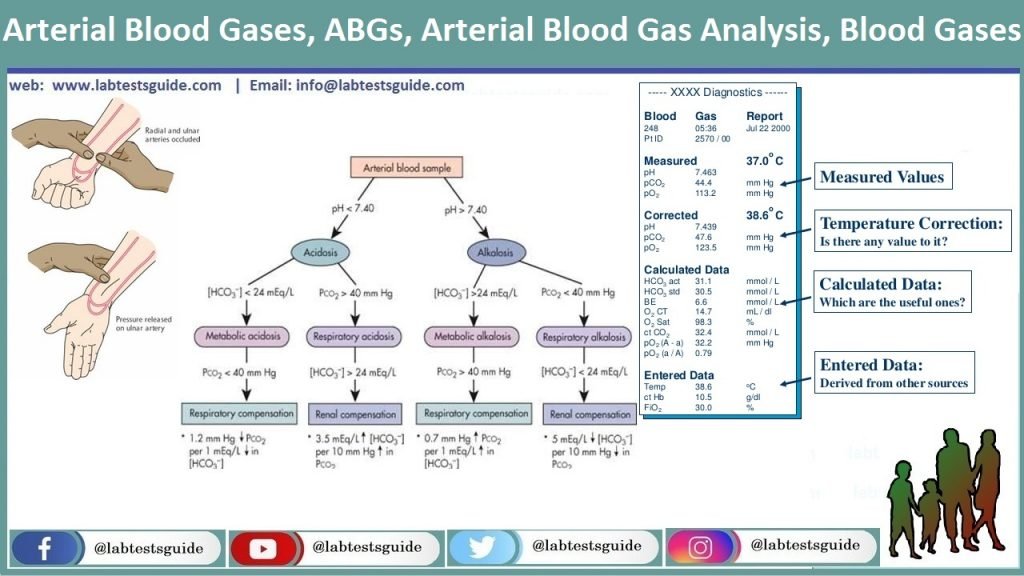
The Practitioner's Acid–Base Primer: Obtaining & Interpreting Blood Gases | Today's Veterinary Practice

DEFINITIONS AND TERMINOLOGY IN BLOOD ACID‐BASE CHEMISTRY - Astrup - 1966 - Annals of the New York Academy of Sciences - Wiley Online Library




![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/3-TableI-1.png)



![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/6-TableIV-1.png)









![PDF] Base Excess : the basics October 2017 | Semantic Scholar PDF] Base Excess : the basics October 2017 | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/6f8a9a879c3efdd0170a857700d730c32f8347d2/5-TableIII-1.png)