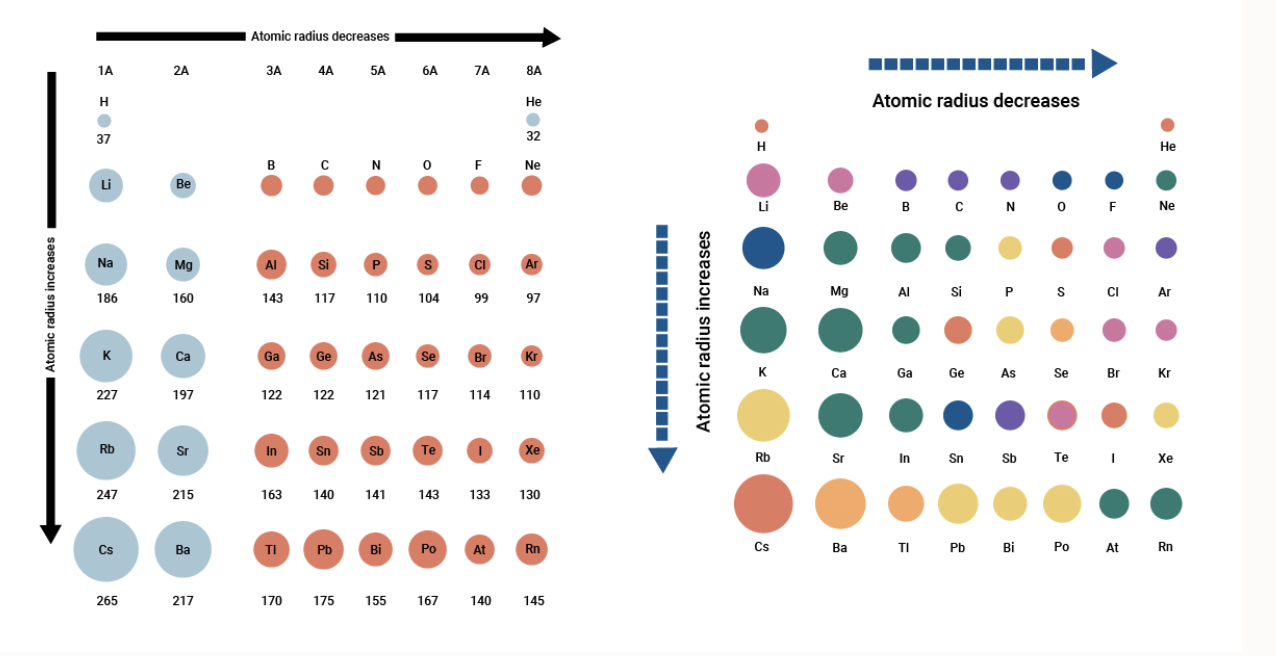
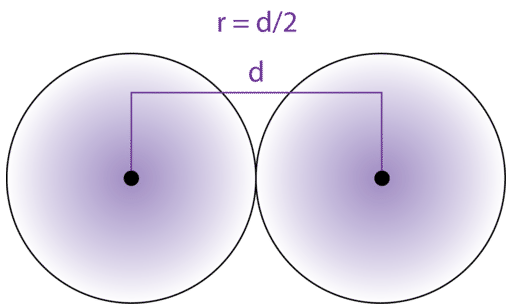
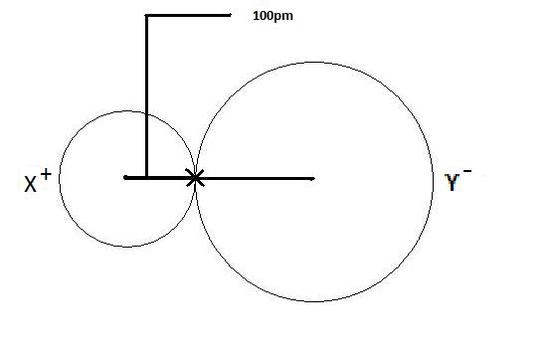
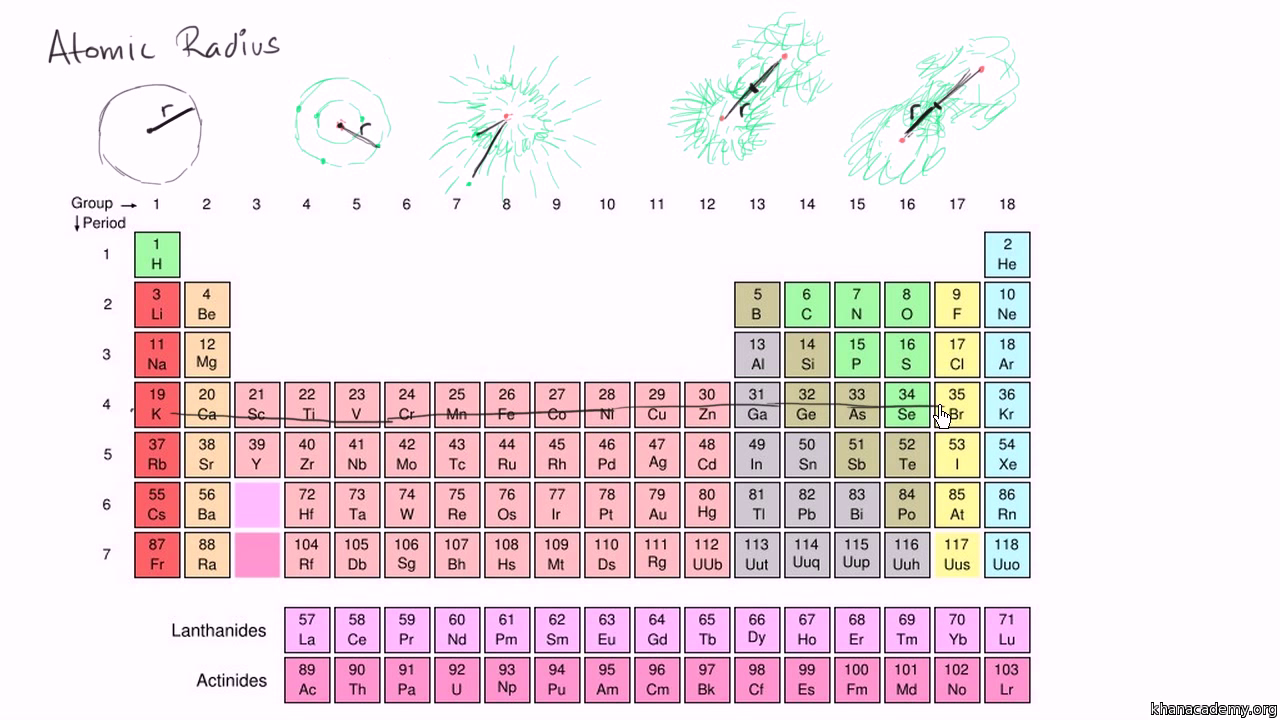
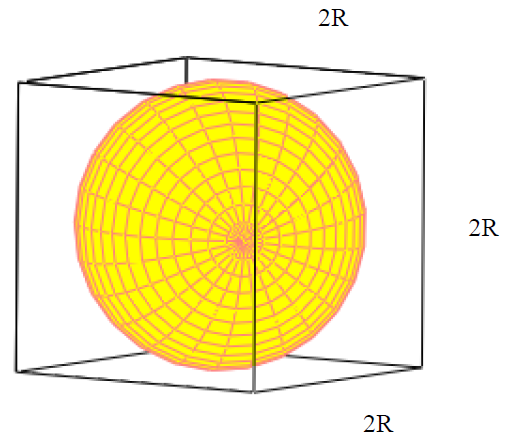
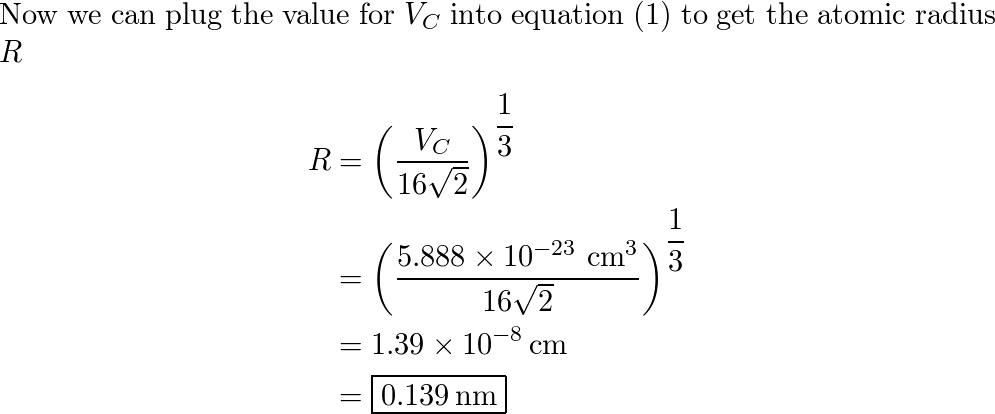Calculate the atomic radius of elementary silver which crystallises in face centred cubic lattice with unit cell edge length 4.086 xx 10^(-10) m.
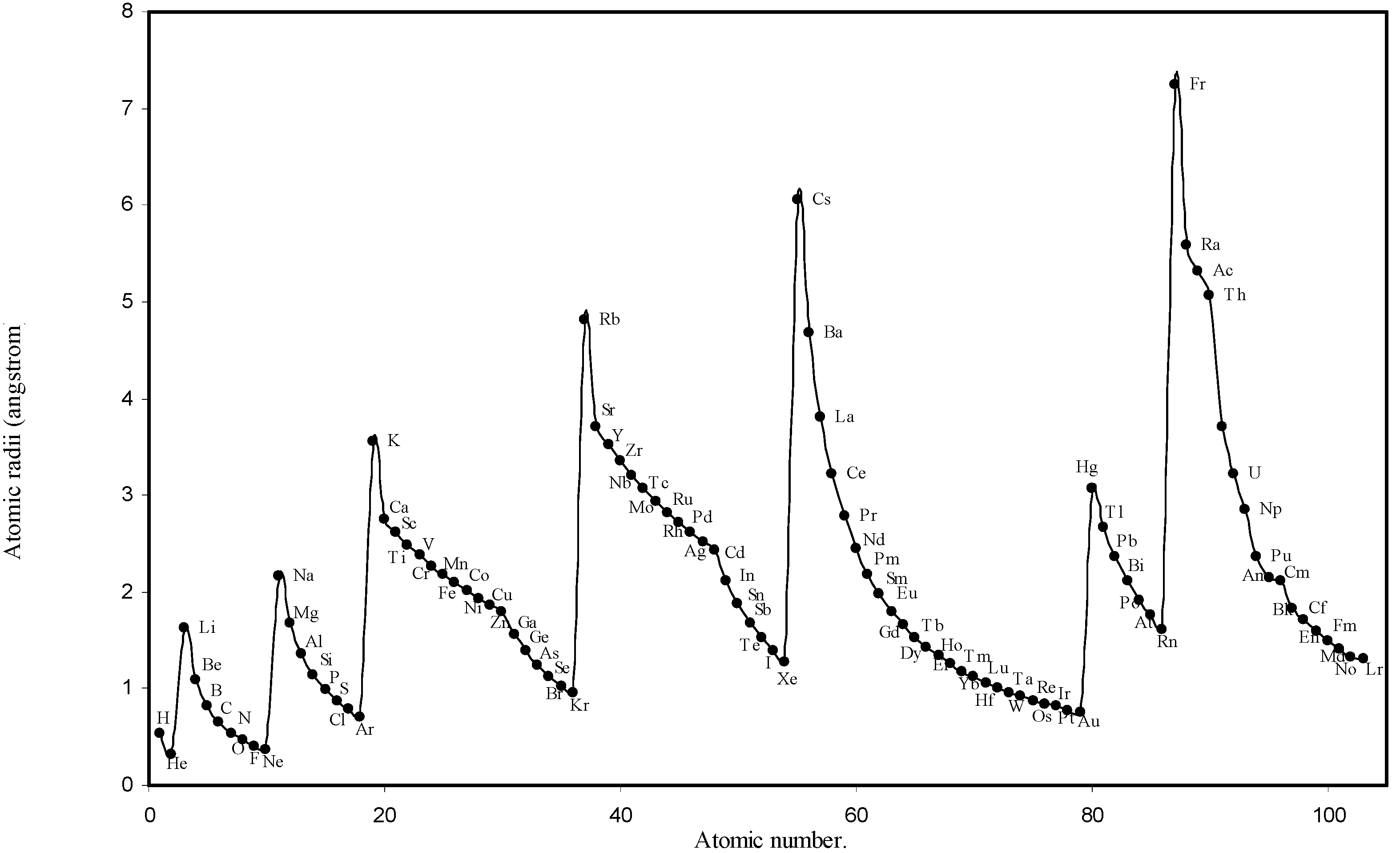
IJMS | Free Full-Text | Theoretical Calculation of Absolute Radii of Atoms and Ions. Part 1. The Atomic Radii

Niobium has a density of 8.57 g/cm3 and crystallizes with the body-centered cubic unit cell. Calculate the radius of a niobium atom - Chemistry Stack Exchange