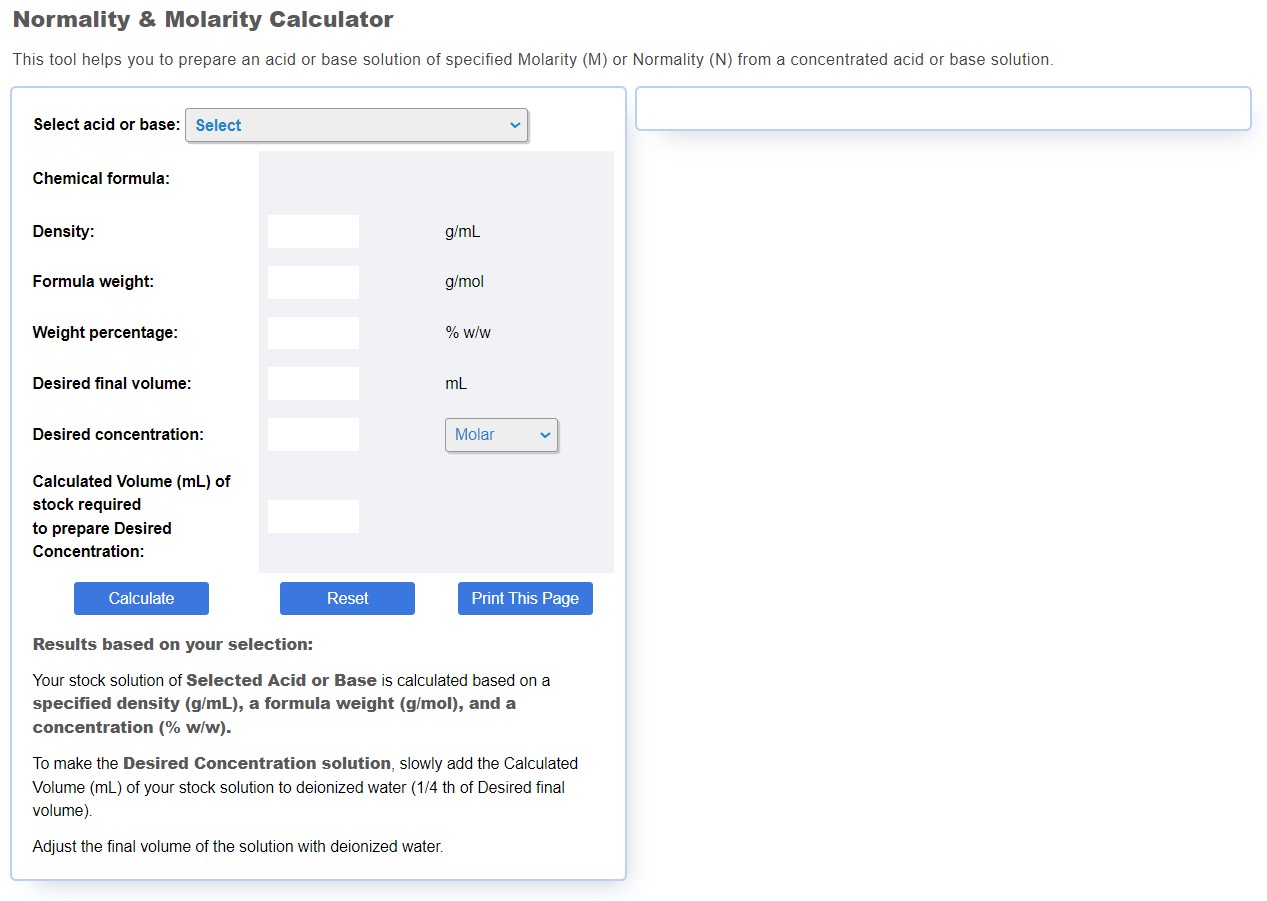
Calculate the normality of a solution containing 15.8 g of `KMnO_(4)` in 50 mL acidic solution. - YouTube

Calculate the molarity `(M)` and normality `(N)` of a solution of oxalic acid `[(COOH)_(2) . 2H_... - YouTube

Calculate the molarity `(M)` and normality `(N)` of a solution of oxalic acid `[(COOH)_(2) . - YouTube
![Calculate the molarity (M) and normality (N) of a solution of oxalic acid [(COOH)(2) . 2H(2) O] containing 12.6 g of the acid in 500 mL of the solution. Calculate the molarity (M) and normality (N) of a solution of oxalic acid [(COOH)(2) . 2H(2) O] containing 12.6 g of the acid in 500 mL of the solution.](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/10487705_web.png)



![Normality - Formula, Definition, Calculations [Solved Examples] Normality - Formula, Definition, Calculations [Solved Examples]](https://i.ytimg.com/vi/XjeaQzl_Wr8/sddefault.jpg)






![Normality - Formula, Definition, Calculations [Solved Examples] - Edureify-Blog Normality - Formula, Definition, Calculations [Solved Examples] - Edureify-Blog](https://notes.edureify.com/wp-content/uploads/2022/05/Blue-Feminine-Plus-Size-Models-In-Trendy-Clothes-Plus-Size-Collection-Banner-93.png)