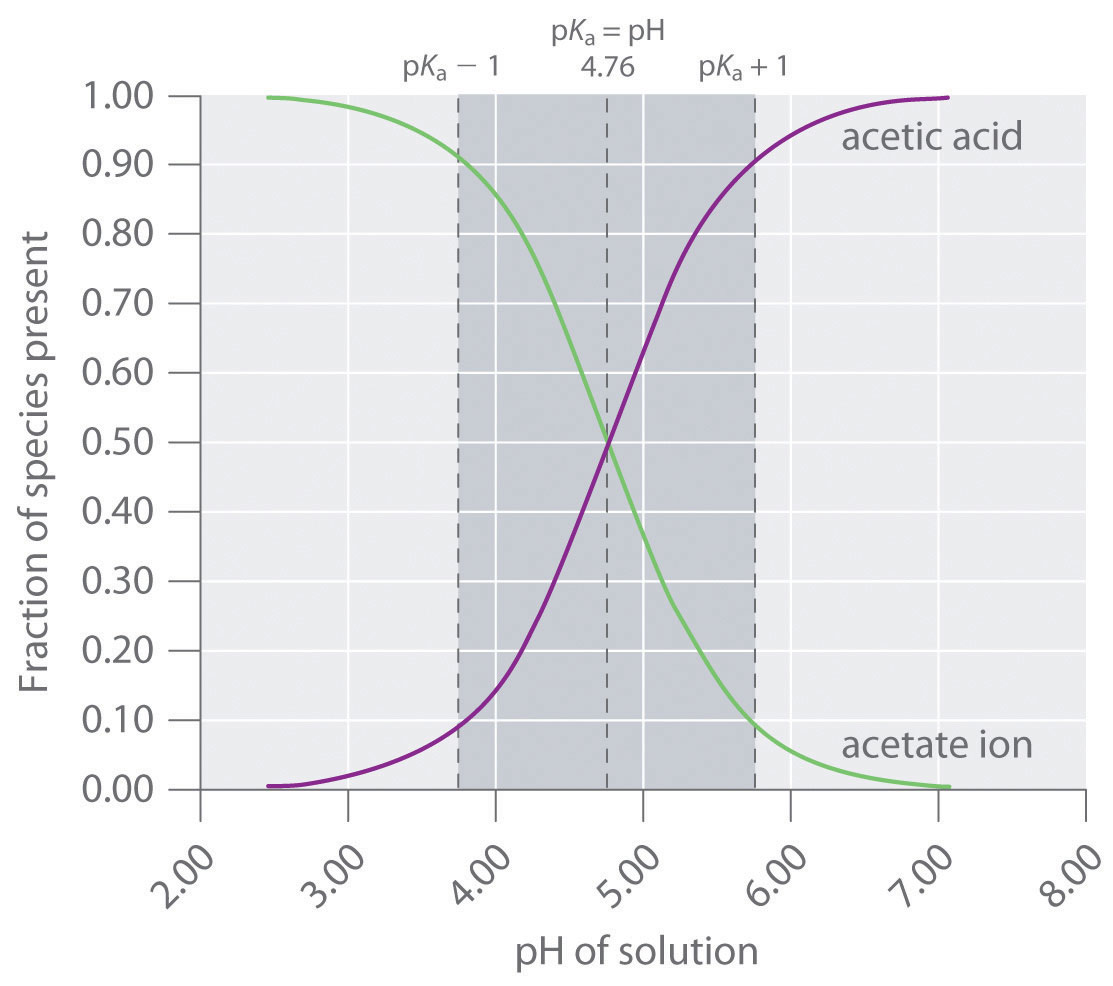
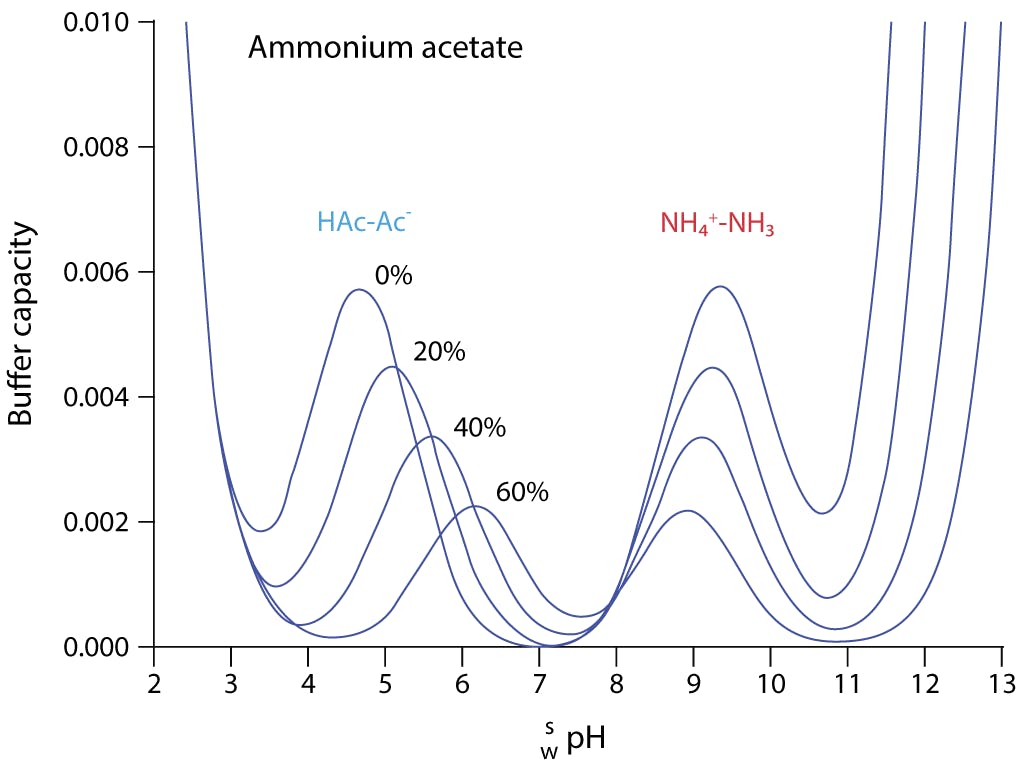
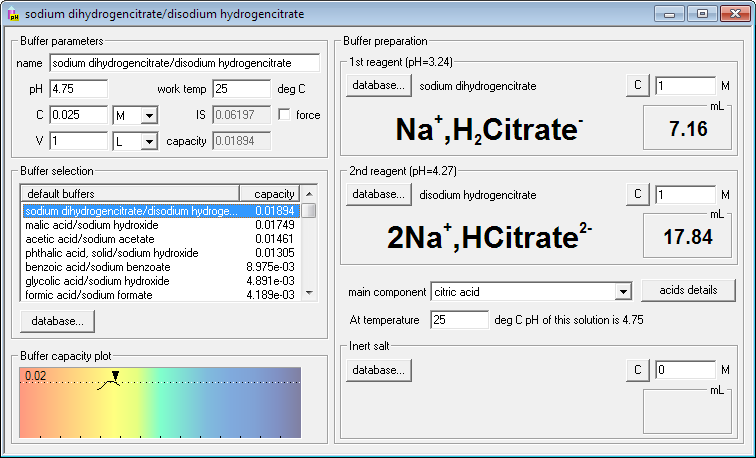
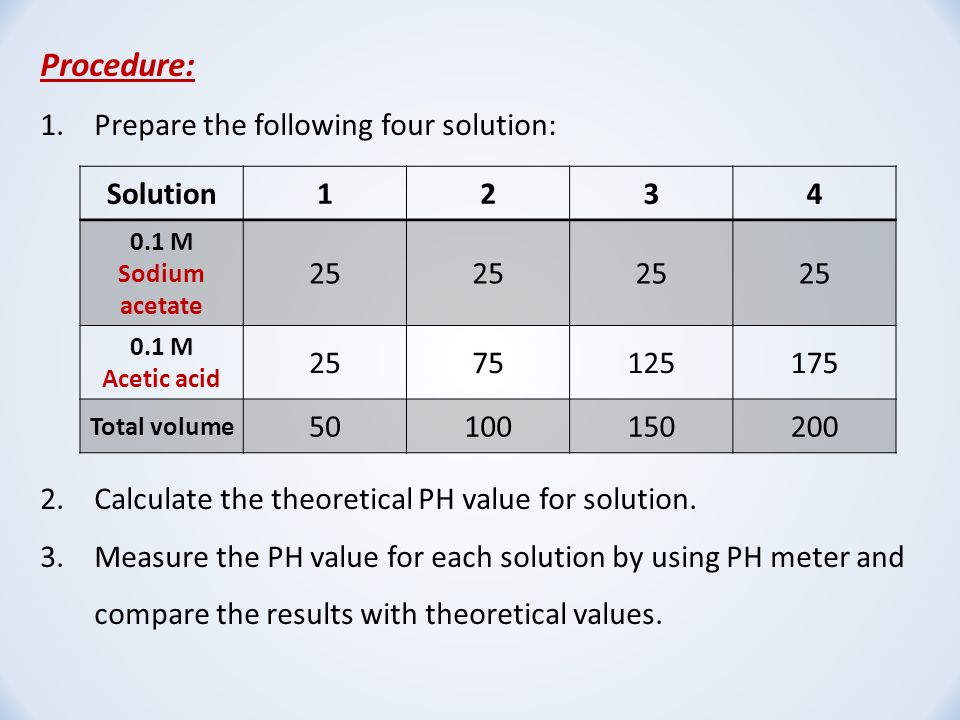
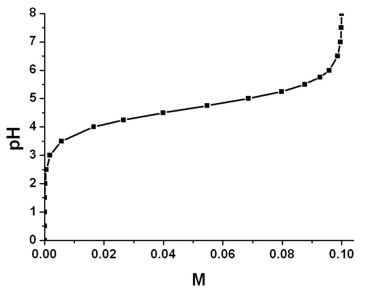
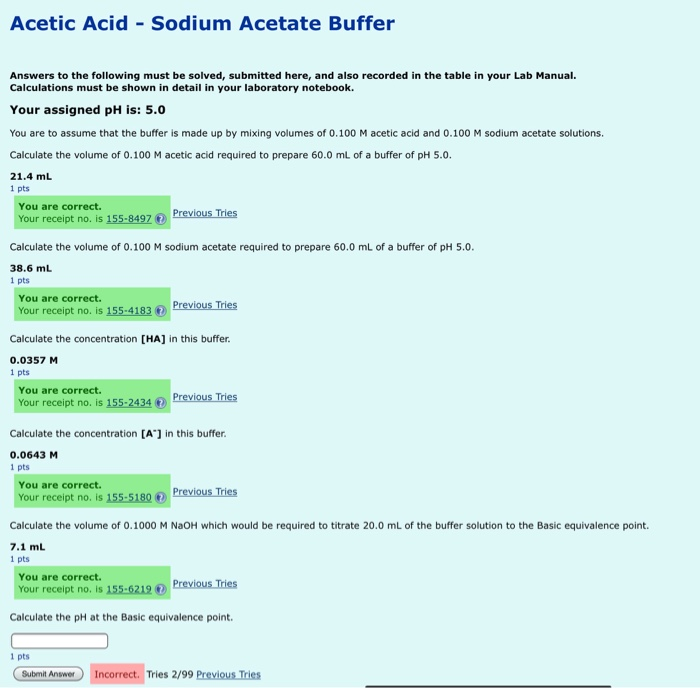
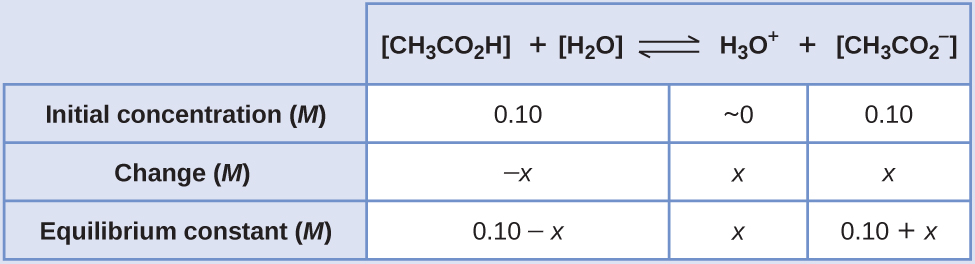
SOLVED: Using Table calculate the expected pH 0f a 0.200 M acetate mixing 50.00 mL 0i 0.200 M acetic acid and 40.00 mL of 0, buffer prepared by does the calculated value

SOLVED: 1. To prepare 0.05 Liter of acetic acid-sodium acetate buffer solution (1 Molar, ph 5.6), what should be the mass in grams of sodium acetate? 2. To prepare 0.05 Liter of

Acid-Base Buffers Equation & Examples | How to Calculate pH of a Buffer - Video & Lesson Transcript | Study.com

pH calculations and more in fundamentals of pharmaceutics. : Calculate pH of 100 ml buffer solution containing 0.1 g acetic acid and 0.2 g sodium actetate.

![BS093] 1.5M Sodium Acetate, pH 6.5 | Biosolution BS093] 1.5M Sodium Acetate, pH 6.5 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2016/02/BS005-Sodium-Acetate-Solution.jpg)
![Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes Sodium Acetate [CH3COONa] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2021/11/sodium-acetate-molecular-weight-calculation.jpg)